Page 99 - Read Online
P. 99
Page 6 of 18 Yang et al. Energy Mater 2023;3:300029 https://dx.doi.org/10.20517/energymater.2023.10
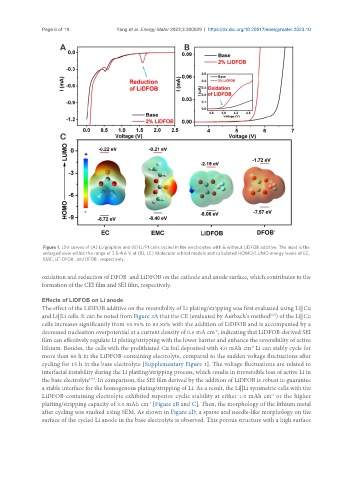
Figure 1. LSV curves of (A) Li/graphite and (B) Li/Pt cells cycled in the electrolytes with & without LiDFOB additive. The inset is the
enlarged view within the range of 3.5-4.6 V of (B); (C) Molecular orbital models and calculated HOMO/LUMO energy levels of EC,
-
+
-
EMC, Li -DFOB , and DFOB , respectively.
oxidation and reduction of DFOB and LiDFOB on the cathode and anode surface, which contributes to the
-
formation of the CEI film and SEI film, respectively.
Effects of LiDFOB on Li anode
The effect of the LiDFOB additive on the reversibility of Li plating/stripping was first evaluated using Li||Cu
and Li||Li cells. It can be noted from Figure 2A that the CE (evaluated by Aurbach’s method ) of the Li||Cu
[65]
cells increases significantly from 53.95% to 83.82% with the addition of LiDFOB and is accompanied by a
decreased nucleation overpotential at a current density of 0.5 mA cm , indicating that LiDFOB-derived SEI
-2
film can effectively regulate Li plating/stripping with the lower barrier and enhance the reversibility of active
lithium. Besides, the cells with the prelithiated Cu foil deposited with 4.0 mAh cm Li can stably cycle for
-2
more than 60 h in the LiDFOB-containing electrolyte, compared to the sudden voltage fluctuations after
cycling for 15 h in the base electrolyte [Supplementary Figure 1]. The voltage fluctuations are related to
interfacial instability during the Li platting/stripping process, which results in irreversible loss of active Li in
the base electrolyte . In comparison, the SEI film derived by the addition of LiDFOB is robust to guarantee
[65]
a stable interface for the homogenous plating/stripping of Li. As a result, the Li||Li symmetric cells with the
-2
LiDFOB-containing electrolyte exhibited superior cyclic stability at either 1.0 mAh cm or the higher
platting/stripping capacity of 3.0 mAh cm [Figure 2B and C]. Then, the morphology of the lithium metal
-2
after cycling was studied using SEM. As shown in Figure 2D, a sparse and needle-like morphology on the
surface of the cycled Li anode in the base electrolyte is observed. This porous structure with a high surface