Page 59 - Read Online
P. 59
Page 10 of 30 Mazzapioda et al. Energy Mater 2023;3:300019 https://dx.doi.org/10.20517/energymater.2023.03
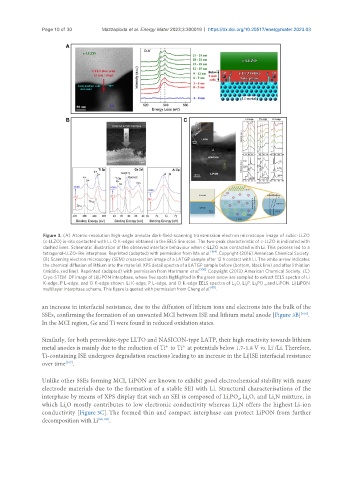
Figure 3. (A) Atomic-resolution high-angle annular dark-field-scanning transmission electron microscope image of cubic-LLZO
(c-LLZO) in-situ contacted with Li. O K-edges obtained in the EELS line scan. The two-peak characteristic of c-LLZO is indicated with
dashed lines. Schematic illustration of the observed interface behaviour when c-LLZO was contacted with Li. This process led to a
tetragonal-LLZO-like interphase. Reprinted (adapted) with permission from Ma et al. [104] . Copyright (2016) American Chemical Society.
(B) Scanning electron microscopy (SEM) cross-section image of a LATGP sample after 12 h contact with Li. The white arrow indicates
the chemical diffusion of lithium into the material. XPS detail spectra of a LATGP sample before (bottom, black line) and after lithiation
(middle, red line). Reprinted (adapted) with permission from Hartmann et al. [106] . Copyright (2013) American Chemical Society. (C)
Cryo-STEM DF image of Li|LiPON interphase, where five spots highlighted in the green arrow are sampled to extract EELS spectra of Li
K-edge, P L-edge, and O K-edge shown. Li K-edge, P L-edge, and O K-edge EELS spectra of L O, Li P, Li PO , and LiPON. Li|LiPON
4
i2
3
3
multilayer interphase scheme. This figure is quoted with permission from Cheng et al. [68] .
an increase in interfacial resistance, due to the diffusion of lithium ions and electrons into the bulk of the
SSEs, confirming the formation of an unwanted MCI between ISE and lithium metal anode [Figure 3B] .
[106]
In the MCI region, Ge and Ti were found in reduced oxidation states.
Similarly, for both perovskite-type LLTO and NASICON-type LATP, their high reactivity towards lithium
4+
+
metal anodes is mainly due to the reduction of Ti to Ti at potentials below 1.7-1.8 V vs. Li /Li. Therefore,
3+
Ti-containing ISE undergoes degradation reactions leading to an increase in the Li|ISE interfacial resistance
[107]
over time .
Unlike other SSEs forming MCI, LiPON are known to exhibit good electrochemical stability with many
electrode materials due to the formation of a stable SEI with Li. Structural characterisations of the
interphase by means of XPS display that such an SEI is composed of Li PO , Li O, and Li N mixture, in
2
4
3
3
which Li O mostly contributes to low electronic conductivity whereas Li N offers the highest Li-ion
2
3
conductivity [Figure 3C]. The formed thin and compact interphase can protect LiPON from further
decomposition with Li [68,108] .