Page 54 - Read Online
P. 54
Mazzapioda et al. Energy Mater 2023;3:300019 https://dx.doi.org/10.20517/energymater.2023.03 Page 5 of 30
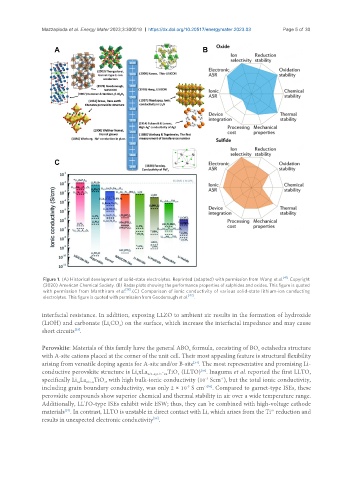
Figure 1. (A) Historical development of solid-state electrolytes. Reprinted (adapted) with permission from Wang et al. [41] . Copyright
(2020) American Chemical Society. (B) Radar plots showing the performance properties of sulphides and oxides. This figure is quoted
with permission from Manthiram et al. [18] . (C) Comparison of ionic conductivity of various solid-state lithium-ion conducting
electrolytes. This figure is quoted with permission from Goodenough et al. [42] .
interfacial resistance. In addition, exposing LLZO to ambient air results in the formation of hydroxide
(LiOH) and carbonate (Li CO ) on the surface, which increase the interfacial impedance and may cause
3
2
[53]
short circuits .
Perovskite: Materials of this family have the general ABO formula, consisting of BO octahedra structure
3
6
with A-site cations placed at the corner of the unit cell. Their most appealing feature is structural flexibility
arising from versatile doping agents for A-site and/or B-site . The most representative and promising Li-
[54]
conductive perovskite structure is Li xLa 2/3-xy1/3 2x 3 [55]
- TiO (LLTO) . Inaguma et al. reported the first LLTO,
3
-3
-1
specifically Li La 2/3-x TiO , with high bulk-ionic conductivity (10 Scm ), but the total ionic conductivity,
3x
3
including grain boundary conductivity, was only 2 × 10 S cm -1[56] . Compared to garnet-type ISEs, these
-5
perovskite compounds show superior chemical and thermal stability in air over a wide temperature range.
Additionally, LLTO-type ISEs exhibit wide ESW; thus, they can be combined with high-voltage cathode
materials . In contrast, LLTO is unstable in direct contact with Li, which arises from the Ti reduction and
4+
[57]
results in unexpected electronic conductivity .
[58]