Page 199 - Read Online
P. 199
Page 10 of 23 Yang et al. Energy Mater 2024;4:400061 https://dx.doi.org/10.20517/energymater.2023.144
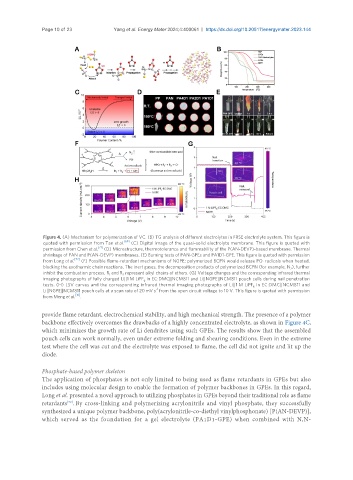
Figure 4. (A) Mechanism for polymerization of VC. (B) TG analysis of different electrolytes in FRSE electrolyte system. This figure is
quoted with permission from Tan et al. [69] (C) Digital image of the quasi-solid electrolyte membrane. This figure is quoted with
permission from Chen et al. [71] (D) Microstructure, thermotolerance and flammability of the P(AN-DEVP)-based membrane. Thermal
shrinkage of PAN and P(AN-DEVP) membranes. (E) Burning tests of PAN-GPEs and PA1D1-GPE. This figure is quoted with permission
from Long et al. [72] (F) Possible flame-retardant mechanisms of NGPE; polymerized BCPN would release PO· radicals when heated,
blocking the exothermic chain reactions. The inert gases, the decomposition products of polymerized BCPN (for example, N ), further
2
inhibit the combustion process. R and R represent alkyl chains of ethers. (G) Voltage changes and the corresponding infrared thermal
1
2
imaging photographs of fully charged Li||1 M LiPF in EC:DMC||NCM811 and Li||NGPE||NCM811 pouch cells during nail penetration
6
tests. (H) LSV curves and the corresponding infrared thermal imaging photographs of Li||1 M LiPF in EC:DMC||NCM811 and
6
-1
Li||NGPE||NCM811 pouch cells at a scan rate of 20 mV s from the open circuit voltage to 10 V. This figure is quoted with permission
from Meng et al. [16] .
provide flame retardant, electrochemical stability, and high mechanical strength. The presence of a polymer
backbone effectively overcomes the drawbacks of a highly concentrated electrolyte, as shown in Figure 4C,
which minimizes the growth rate of Li dendrites using such GPEs. The results show that the assembled
pouch cells can work normally, even under extreme folding and shearing conditions. Even in the extreme
test where the cell was cut and the electrolyte was exposed to flame, the cell did not ignite and lit up the
diode.
Phosphate-based polymer skeleton
The application of phosphates is not only limited to being used as flame retardants in GPEs but also
includes using molecular design to enable the formation of polymer backbones in GPEs. In this regard,
Long et al. presented a novel approach to utilizing phosphates in GPEs beyond their traditional role as flame
retardants . By cross-linking and polymerizing acrylonitrile and vinyl phosphate, they successfully
[72]
synthesized a unique polymer backbone, poly(acrylonitrile-co-diethyl vinylphosphonate) [P(AN-DEVP)],
which served as the foundation for a gel electrolyte (PA1D1-GPE) when combined with N,N-