Page 204 - Read Online
P. 204
Yang et al. Energy Mater 2024;4:400061 https://dx.doi.org/10.20517/energymater.2023.144 Page 15 of 23
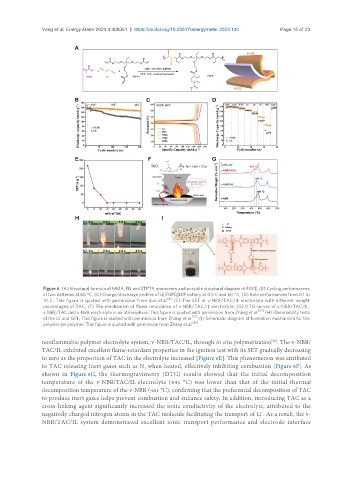
Figure 6. (A) Structural formula of MMA, FN and ETPTA monomers and possible structural diagram of FGPE; (B) Cycling performances
of two batteries at 60 °C; (C) Charge/discharge profiles of Li||FGPE||LFP battery at 0.5 C and 60 °C; (D) Rate performances from 0.1 to
[81]
10 C; This figure is quoted with permission from Sun et al. (E) The SET of v-NBR/TAC/IL electrolyte with different weight
percentages of TAC; (F) The mechanism of flame retardance of v-NBR/TAC/IL electrolyte; (G) DTG curves of v-NBR/TAC/IL,
[82]
v-NBR/TAC and v-NBR electrolyte in air atmosphere; This figure is quoted with permission from Zhang et al. (H) Flammability tests
[57]
of the LE and GPE; This figure is quoted with permission from Zhang et al. ; (I) Schematic diagram of formation mechanism for the
[85]
polymer gel polymer; This figure is quoted with permission from Zhang et al. .
nonflammable polymer electrolyte system, v-NBR/TAC/IL, through in situ polymerization . The v-NBR/
[82]
TAC/IL exhibited excellent flame-retardant properties in the ignition test with its SET gradually decreasing
to zero as the proportion of TAC in the electrolyte increased [Figure 6E]. This phenomenon was attributed
to TAC releasing inert gases such as N when heated, effectively inhibiting combustion [Figure 6F]. As
2
shown in Figure 6G, the thermogravimetry (DTG) results showed that the initial decomposition
temperature of the v-NBR/TAC/IL electrolyte (441 °C) was lower than that of the initial thermal
decomposition temperature of the v-NBR (481 °C), confirming that the preferential decomposition of TAC
to produce inert gases helps prevent combustion and enhance safety. In addition, introducing TAC as a
cross-linking agent significantly increased the ionic conductivity of the electrolyte, attributed to the
negatively charged nitrogen atoms in the TAC molecule facilitating the transport of Li . As a result, the v-
+
NBR/TAC/IL system demonstrated excellent ionic transport performance and electrode interface