Page 64 - Read Online
P. 64
Lin et al. Cancer Drug Resist. 2026;9:14 Page 9 of 19
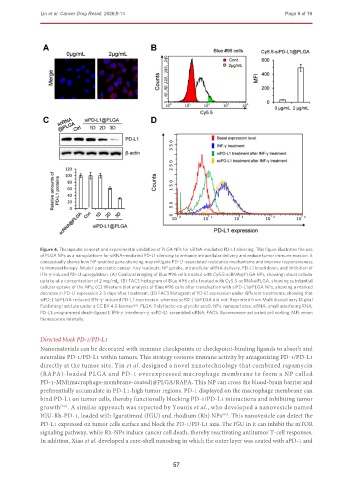
Figure 4. Therapeutic concept and experimental validation of PLGA NPs for siRNA-mediated PD-L1 silencing. This figure illustrates the use
of PLGA NPs as a nanoplatform for siRNA-mediated PD-L1 silencing to enhance intracellular delivery and reduce tumor immune evasion. It
conceptually shows how NP-enabled gene silencing may mitigate PD-L1-associated resistance mechanisms and improve responsiveness
to immunotherapy. Model: pancreatic cancer. Key readouts: NP uptake, intracellular siRNA delivery, PD-L1 knockdown, and inhibition of
IFN-γ-induced PD-L1 upregulation. (A) Confocal imaging of Blue #96 cells treated with Cy5.5-scRNA@PLGA NPs, showing robust cellular
uptake at a concentration of 2 mg/mL; (B) FACS histogram of Blue #96 cells treated with Cy5.5-scRNA@PLGA, showing substantial
cellular uptake of the NPs; (C) Western blot analysis of Blue #96 cells after transfection with siPD-L1@PLGA NPs, showing a marked
decrease in PD-L1 expression 2-3 days after treatment; (D) FACS histogram of PD-L1 expression under different treatments, showing that
siPD-L1@PLGA reduced IFN-γ-induced PD-L1 expression, whereas scPD-L1@PLGA did not. Reprinted from Multidisciplinary Digital
Publishing Institute under a CC BY 4.0 license [64] . PLGA: Poly(lactic-co-glycolic acid); NPs: nanoparticles; siRNA: small interfering RNA;
PD-L1: programmed death-ligand 1; IFN-γ: interferon-γ; scPD-L1: scrambled siRNA; FACS: fluorescence-activated cell sorting; MFI: mean
fluorescence intensity.
Directed block PD-1/PD-L1
Nanomaterials can be decorated with immune checkpoints or checkpoint-binding ligands to absorb and
neutralize PD-1/PD-L1 within tumors. This strategy restores immune activity by antagonizing PD-1/PD-L1
directly at the tumor site. Yin et al. designed a novel nanotechnology that combined rapamycin
(RAPA)-loaded PLGA and PD-1 overexpressed macrophage membrane to form a NP called
PD-1-MM(macrophage-membrane-coated)@PLGA/RAPA. This NP can cross the blood-brain barrier and
preferentially accumulate in PD-L1-high tumor regions. PD-1 displayed on the macrophage membrane can
bind PD-L1 on tumor cells, thereby functionally blocking PD-1/PD-L1 interactions and inhibiting tumor
growth . A similar approach was reported by Younis et al., who developed a nanovesicle named
[68]
IGU-Rh-PD-1, loaded with Iguratimod (IGU) and rhodium (Rh) NPs . This nanovesicle can detect the
[69]
PD-L1 expressed on tumor cells surface and block the PD-1/PD-L1 axis. The IGU in it can inhibit the mTOR
signaling pathway, while Rh-NPs induce cancer cell death, thereby reactivating antitumor T-cell responses.
In addition, Xiao et al. developed a core-shell nanodrug in which the outer layer was coated with aPD-1 and
57