Page 63 - Read Online
P. 63
Page 8 of 19 Lin et al. Cancer Drug Resist. 2026;9:14
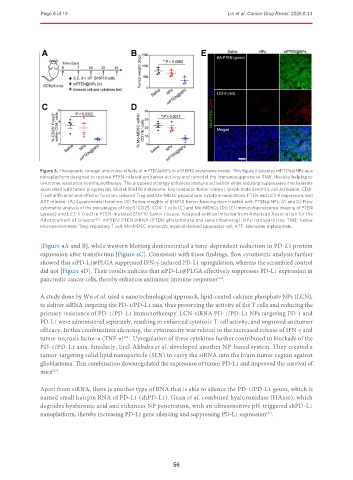
Figure 3. Therapeutic concept and in vivo effects of mPTEN@NPs in a B16F10 melanoma model. This figure illustrates mPTEN@NPs as a
nanoplatform designed to restore PTEN-related antitumor activity and remodel the immunosuppressive TME, thereby helping to
overcome resistance to immunotherapy. The proposed strategy enhances immune activation while reducing suppressive mechanisms
associated with tumor progression. Model: B16F10 melanoma. Key readouts: tumor control, lymph node dendritic cell activation, CD8 +
T-cell infiltration and effector function, reduced Treg and Mo-MDSC populations, cytokine modulation, PTEN and LC3-II expression, and
ATP release. (A) Experimental timeline; (B) Tumor weights of B16F10 tumor-bearing mice treated with PTEN@NPs; (C and D) Flow
cytometric analysis of the percentages of Foxp3 CD25 CD4 T cells (C) and Mo-MDSCs (D); (E) Immunofluorescence imaging of PTEN
+
+
+
(green) and LC3-II (red) in PTEN-mutated B16F10 tumor tissues. Adapted with permission from American Association for the
Advancement of Science [62] . mPTEN: PTEN mRNA (PTEN: phosphatase and tensin homolog); NPs: nanoparticles; TME: tumor
microenvironment; Treg: regulatory T cell; Mo-MDSC: monocytic myeloid-derived suppressor cell; ATP: adenosine triphosphate.
[Figure 4A and B], while western blotting demonstrated a time-dependent reduction in PD-L1 protein
expression after transfection [Figure 4C]. Consistent with these findings, flow cytometric analysis further
showed that siPD-L1@PLGA suppressed IFN-γ-induced PD-L1 upregulation, whereas the scrambled control
did not [Figure 4D]. Their results indicate that siPD-L1@PLGA effectively suppresses PD-L1 expression in
pancreatic cancer cells, thereby enhances antitumor immune responses .
[64]
A study done by Wu et al. used a nanotechnological approach, lipid-coated calcium phosphate NPs (LCN),
to deliver siRNA targeting the PD-1/PD-L1 axis, thus preserving the activity of the T cells and reducing the
primary resistance of PD-1/PD-L1 immunotherapy. LCN-siRNA PD-1/PD-L1 NPs targeting PD-1 and
PD-L1 were administered separately, resulting in enhanced cytotoxic T-cell activity, and improved antitumor
efficacy. In this combination silencing, the cytotoxicity was related to the increased release of IFN-γ and
tumor necrosis factor-α (TNF-α) . Upregulation of these cytokines further contributed to blockade of the
[65]
PD-1/PD-L1 axis. Similarly, Erel-Akbaba et al. developed another NP-based system. They created a
tumor-targeting solid lipid nanoparticle (SLN) to carry the siRNA into the brain tumor region against
glioblastoma. This combination downregulated the expression of tumor PD-L1 and improved the survival of
mice .
[66]
Apart from siRNA, there is another type of RNA that is able to silence the PD-1/PD-L1 genes, which is
named small hairpin RNA of PD-L1 (shPD-L1). Guan et al. combined hyaluronidase (HAase), which
degrades hyaluronic acid and enhances NP penetration, with an ultrasensitive pH-triggered shPD-L1
nanoplatform, thereby increasing PD-L1 gene silencing and suppressing PD-L1 expression .
[67]
56