Page 65 - Read Online
P. 65
Page 10 of 19 Lin et al. Cancer Drug Resist. 2026;9:14
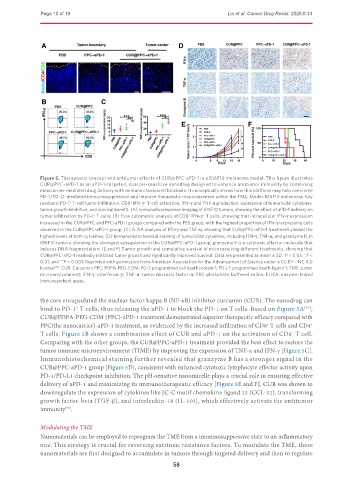
Figure 5. Therapeutic concept and antitumor effects of CUR@PPC-aPD-1 in a B16F10 melanoma model. This figure illustrates
CUR@PPC-aPD-1 as an aPD-1-targeted, dual pH-sensitive nanodrug designed to enhance antitumor immunity by combining
nanocarrier-mediated drug delivery with immune checkpoint blockade. It conceptually shows how this platform may help overcome
PD-1/PD-L1-mediated immunosuppression and improve therapeutic responsiveness within the TME. Model: B16F10 melanoma. Key
readouts: PD-1 T-cell tumor infiltration, CD8 IFN-γ T-cell activation, IFN-γ and TNF-α production, expression of tumoricidal cytokines,
+
+
+
tumor growth inhibition, and survival benefit. (A) Immunofluorescence imaging of B16F10 tumors, showing the effect of aPD-1 delivery on
tumor infiltration by PD-1 T cells; (B) Flow cytometric analysis of CD8 IFN-γ T cells, showing that intracellular IFN-γ expression
+
+
+
increased in the CUR@PPC and PPC-aPD-1 groups compared with the PBS group, with the highest proportion of IFN-γ-expressing cells
observed in the CUR@PPC-aPD-1 group; (C) ELISA analysis of IFN-γ and TNF-α, showing that CUR@PPC-aPD-1 treatment yielded the
highest levels of both cytokines; (D) Immunohistochemical staining of tumoricidal cytokines, including IFN-γ, TNF-α, and granzyme B, in
B16F10 tumors, showing the strongest upregulation in the CUR@PPC-aPD-1 group; granzyme B is a cytotoxic effector molecule that
induces DNA fragmentation; (E and F) Tumor growth and cumulative survival of mice receiving different treatments, showing that
CUR@PPC-aPD-1 markedly inhibited tumor growth and significantly improved survival. Data are presented as mean ± SD. P < 0.05, P <
**
*
0.01, and P < 0.001. Reprinted with permission from American Association for the Advancement of Science under a CC BY - NC 4.0
***
license [70] . CUR: Curcumin; PPC: PDPA-PEG-CDM; PD-1: programmed cell death protein 1; PD-L1: programmed death-ligand 1; TME: tumor
microenvironment; IFN-γ: interferon-γ; TNF-α: tumor necrosis factor-α; PBS: phosphate-buffered saline; ELISA: enzyme-linked
immunosorbent assay.
the core encapsulated the nuclear factor kappa B (NF-κB) inhibitor curcumin (CUR). The nanodrug can
bind to PD-1 T cells, thus releasing the aPD-1 to block the PD-1 on T cells. Based on Figure 5A ,
[70]
+
CUR@PDPA-PEG-CDM (PPC)-aPD-1 treatment demonstrated superior therapeutic efficacy compared with
PPC(the nanocarrier)-aPD-1 treatment, as evidenced by the increased infiltration of CD8 T cells and CD4 +
+
T cells. Figure 5B shows a combination effect of CUR and aPD-1 on the activation of CD8 T cell.
+
Comparing with the other groups, the CUR@PPC-aPD-1 treatment provided the best effect to restore the
tumor immune microenvironment (TIME) by improving the expression of TNF-α and IFN-γ [Figure 5C].
Immunohistochemical staining further revealed that granzyme B has a stronger signal in the
CUR@PPC-aPD-1 group [Figure 5D], consistent with enhanced cytotoxic lymphocyte effector activity upon
PD-1/PD-L1 checkpoint inhibition. The pH-sensitive nanomicelle plays a crucial role in ensuring effective
delivery of aPD-1 and maximizing its immunotherapeutic efficacy [Figure 5E and F]. CUR was shown to
downregulate the expression of cytokines like [C-C motif chemokine ligand 22 (CCL-22), transforming
growth factor-beta (TGF-β), and interleukin-10 (IL-10)], which effectively activate the antitumor
immunity .
[70]
Modulating the TME
Nanomaterials can be employed to reprogram the TME from a immunosuppressive state to an inflammatory
one. This strategy is crucial for reversing extrinsic resistance factors. To modulate the TME, these
nanomaterials are first designed to accumulate in tumors through targeted delivery and then to regulate
58