Page 807 - Read Online
P. 807
Tredway et al. Mini-invasive Surg 2020;4:78 I http://dx.doi.org/10.20517/2574-1225.2020.77 Page 7 of 11
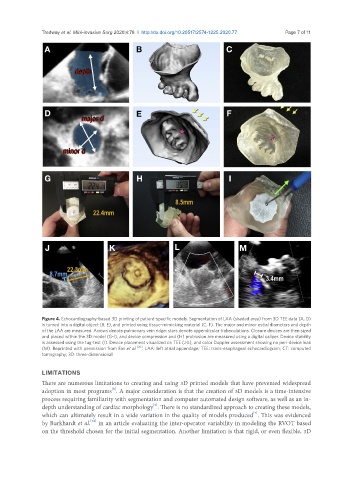
Figure 4. Echocardiography-based 3D printing of patient-specific models. Segmentation of LAA (shaded area) from 3D TEE data (A, D)
is turned into a digital object (B, E), and printed using tissue-mimicking material (C, F). The major and minor ostial diameters and depth
of the LAA are measured. Arrows denote pulmonary vein ridge; stars denote appendicular trabeculations. Closure devices are then sized
and placed within the 3D model (G-I), and device compression and (H) protrusion are measured using a digital caliper. Device stability
is assessed using the tug-test (I). Device placement visualized on TEE (J-L), and color Doppler assessment showing no peri-device leak
(M). Reprinted with permission from Fan et al. [20] . LAA: left atrial appendage; TEE: trans-esophageal echocardiogram; CT: computed
tomography; 3D: three-dimensional
LIMITATIONS
There are numerous limitations to creating and using 3D printed models that have prevented widespread
[8]
adoption in most programs . A major consideration is that the creation of 3D models is a time-intensive
process requiring familiarity with segmentation and computer automated design software, as well as an in-
[5]
depth understanding of cardiac morphology . There is no standardized approach to creating these models,
which can ultimately result in a wide variation in the quality of models produced . This was evidenced
[7]
[24]
by Burkhardt et al. in an article evaluating the inter-operator variability in modeling the RVOT based
on the threshold chosen for the initial segmentation. Another limitation is that rigid, or even flexible, 3D