Page 92 - Read Online
P. 92
Page 8 of 12 Chi et al. J. Mater. Inf. 2025, 5, 11 https://dx.doi.org/10.20517/jmi.2024.49
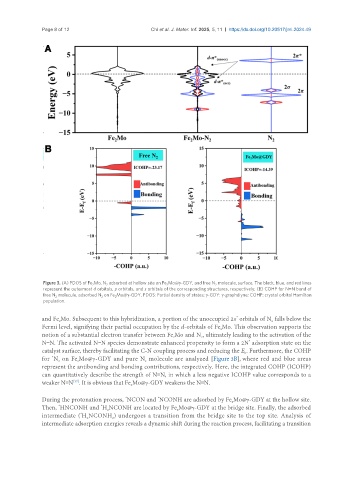
Figure 3. (A) PDOS of Fe Mo, N adsorbed at hollow site on Fe Mo@γ-GDY, and free N molecule, surface. The black, blue, and red lines
2
2
2
2
represent the outermost d orbitals, p orbitals, and s orbitals of the corresponding structures, respectively; (B) COHP for N≡N bond of
free N molecule, adsorbed N on Fe Mo@γ-GDY. PDOS: Partial density of states; γ-GDY: γ-graphdiyne; COHP: crystal orbital Hamilton
2
2
2
population.
and Fe Mo. Subsequent to this hybridization, a portion of the unoccupied 2π orbitals of N falls below the
*
2
2
Fermi level, signifying their partial occupation by the d-orbitals of Fe Mo. This observation supports the
2
notion of a substantial electron transfer between Fe Mo and N , ultimately leading to the activation of the
2
2
N≡N. The activated N≡N species demonstrate enhanced propensity to form a 2N adsorption state on the
*
catalyst surface, thereby facilitating the C-N coupling process and reducing the E . Furthermore, the COHP
b
for N on Fe Mo@γ-GDY and pure N molecule are analyzed [Figure 3B], where red and blue areas
*
2
2
2
represent the antibonding and bonding contributions, respectively. Here, the integrated COHP (ICOHP)
can quantitatively describe the strength of N≡N, in which a less negative ICOHP value corresponds to a
[37]
weaker N≡N . It is obvious that Fe Mo@γ-GDY weakens the N≡N.
2
*
During the protonation process, NCON and NCONH are adsorbed by Fe Mo@γ-GDY at the hollow site.
*
2
Then, HNCONH and H NCONH are located by Fe Mo@γ-GDY at the bridge site. Finally, the adsorbed
*
*
2
2
intermediate ( H NCONH ) undergoes a transition from the bridge site to the top site. Analysis of
*
2
2
intermediate adsorption energies reveals a dynamic shift during the reaction process, facilitating a transition