Page 91 - Read Online
P. 91
Chi et al. J. Mater. Inf. 2025, 5, 11 https://dx.doi.org/10.20517/jmi.2024.49 Page 7 of 12
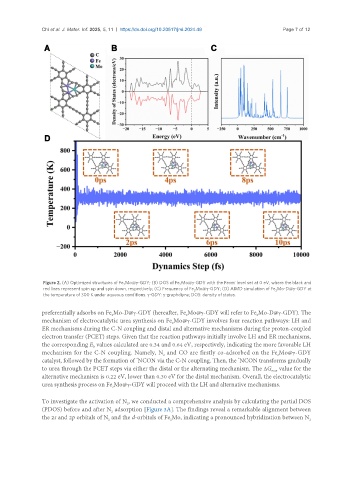
Figure 2. (A) Optimized structures of Fe Mo@γ-GDY; (B) DOS of Fe Mo@γ-GDY with the Fermi level set at 0 eV, where the black and
2 2
red lines represent spin up and spin down, respectively; (C) Frequency of Fe Mo@γ-GDY; (D) AIMD simulation of Fe Mo-D@γ-GDY at
2 2
the temperature of 300 K under aqueous conditions. γ-GDY: γ-graphdiyne; DOS: density of states.
preferentially adsorbs on Fe Mo-D@γ-GDY (hereafter, Fe Mo@γ-GDY will refer to Fe Mo-D@γ-GDY). The
2
2
2
mechanism of electrocatalytic urea synthesis on Fe Mo@γ-GDY involves four reaction pathways: LH and
2
ER mechanisms during the C-N coupling and distal and alternative mechanisms during the proton-coupled
electron transfer (PCET) steps. Given that the reaction pathways initially involve LH and ER mechanisms,
the corresponding E values calculated are 0.34 and 0.64 eV, respectively, indicating the more favorable LH
b
mechanism for the C-N coupling. Namely, N and CO are firstly co-adsorbed on the Fe Mo@γ-GDY
2
2
catalyst, followed by the formation of NCON via the C-N coupling. Then, the NCON transforms gradually
*
*
to urea through the PCET steps via either the distal or the alternating mechanism. The ΔG value for the
max
alternative mechanism is 0.22 eV, lower than 0.30 eV for the distal mechanism. Overall, the electrocatalytic
urea synthesis process on Fe Mo@γ-GDY will proceed with the LH and alternative mechanisms.
2
To investigate the activation of N , we conducted a comprehensive analysis by calculating the partial DOS
2
(PDOS) before and after N adsorption [Figure 3A]. The findings reveal a remarkable alignment between
2
the 2s and 2p orbitals of N and the d-orbitals of Fe Mo, indicating a pronounced hybridization between N
2
2
2