Page 93 - Read Online
P. 93
Chi et al. J. Mater. Inf. 2025, 5, 11 https://dx.doi.org/10.20517/jmi.2024.49 Page 9 of 12
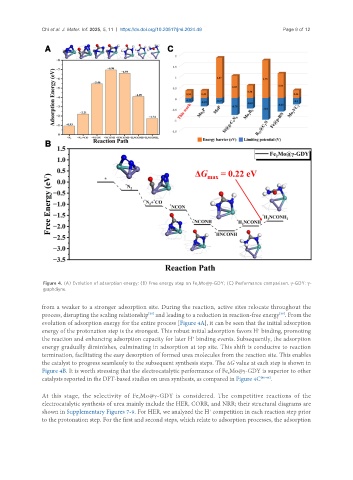
Figure 4. (A) Evolution of adsorption energy; (B) Free energy step on Fe Mo@γ-GDY; (C) Performance comparison. γ-GDY: γ-
2
graphdiyne.
from a weaker to a stronger adsorption site. During the reaction, active sites relocate throughout the
[38]
process, disrupting the scaling relationship and leading to a reduction in reaction-free energy . From the
[39]
evolution of adsorption energy for the entire process [Figure 4A], it can be seen that the initial adsorption
energy of the protonation step is the strongest. This robust initial adsorption favors H binding, promoting
+
+
the reaction and enhancing adsorption capacity for later H binding events. Subsequently, the adsorption
energy gradually diminishes, culminating in adsorption at top site. This shift is conducive to reaction
termination, facilitating the easy desorption of formed urea molecules from the reaction site. This enables
the catalyst to progress seamlessly to the subsequent synthesis steps. The ΔG value at each step is shown in
Figure 4B. It is worth stressing that the electrocatalytic performance of Fe Mo@γ-GDY is superior to other
2
catalysts reported in the DFT-based studies on urea synthesis, as compared in Figure 4C [40-46] .
At this stage, the selectivity of Fe Mo@γ-GDY is considered. The competitive reactions of the
2
electrocatalytic synthesis of urea mainly include the HER, CORR, and NRR; their structural diagrams are
shown in Supplementary Figures 7-9. For HER, we analyzed the H competition in each reaction step prior
+
to the protonation step. For the first and second steps, which relate to adsorption processes, the adsorption