Page 62 - Read Online
P. 62
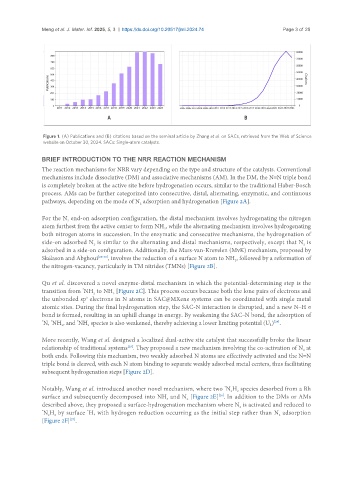
Meng et al. J. Mater. Inf. 2025, 5, 3 https://dx.doi.org/10.20517/jmi.2024.74 Page 3 of 25
Figure 1. (A) Publications and (B) citations based on the seminal article by Zhang et al. on SACs, retrieved from the Web of Science
website on October 30, 2024. SACs: Single-atom catalysts.
BRIEF INTRODUCTION TO THE NRR REACTION MECHANISM
The reaction mechanisms for NRR vary depending on the type and structure of the catalysts. Conventional
mechanisms include dissociative (DM) and associative mechanisms (AM). In the DM, the N≡N triple bond
is completely broken at the active site before hydrogenation occurs, similar to the traditional Haber-Bosch
process. AMs can be further categorized into consecutive, distal, alternating, enzymatic, and continuous
pathways, depending on the mode of N adsorption and hydrogenation [Figure 2A].
2
For the N end-on adsorption configuration, the distal mechanism involves hydrogenating the nitrogen
2
atom furthest from the active center to form NH , while the alternating mechanism involves hydrogenating
3
both nitrogen atoms in succession. In the enzymatic and consecutive mechanisms, the hydrogenation of
side-on adsorbed N is similar to the alternating and distal mechanisms, respectively, except that N is
2
2
adsorbed in a side-on configuration. Additionally, the Mars-van-Krevelen (MvK) mechanism, proposed by
Skúlason and Abghoui [28-30] , involves the reduction of a surface N atom to NH , followed by a reformation of
3
the nitrogen-vacancy, particularly in TM nitrides (TMNs) [Figure 2B].
Qu et al. discovered a novel enzyme-distal mechanism in which the potential-determining step is the
transition from NH to NH [Figure 2C]. This process occurs because both the lone pairs of electrons and
*
3
2
the unbonded sp electrons in N atoms in SAC@MXene systems can be coordinated with single metal
3
atomic sites. During the final hydrogenation step, the SAC-N interaction is disrupted, and a new N–H σ
bond is formed, resulting in an uphill change in energy. By weakening the SAC–N bond, the adsorption of
[24]
*
*
* N, NH , and NH species is also weakened, thereby achieving a lower limiting potential (U ) .
3
L
2
More recently, Wang et al. designed a localized dual-active site catalyst that successfully broke the linear
[25]
relationship of traditional systems . They proposed a new mechanism involving the co-activation of N at
2
both ends. Following this mechanism, two weakly adsorbed N atoms are effectively activated and the N≡N
triple bond is cleaved, with each N atom binding to separate weakly adsorbed metal centers, thus facilitating
subsequent hydrogenation steps [Figure 2D].
*
Notably, Wang et al. introduced another novel mechanism, where two N H species desorbed from a Rh
2
2
surface and subsequently decomposed into NH and N [Figure 2E] . In addition to the DMs or AMs
[26]
3
2
described above, they proposed a surface-hydrogenation mechanism where N is activated and reduced to
2
*
* N H by surface H, with hydrogen reduction occurring as the initial step rather than N adsorption
2
2
2
[Figure 2F] .
[27]