Page 600 - Read Online
P. 600
Page 8 of 13 Jiang et al. Hepatoma Res 2020;6:52 I http://dx.doi.org/10.20517/2394-5079.2020.47
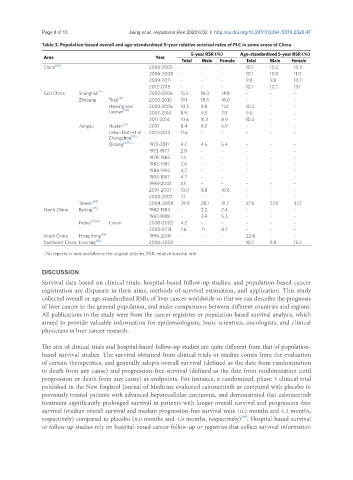
Table 3. Population-based overall and age-standardised 5-year relative survival rates of PLC in some areas of China
5-year RSR (%) Age-standardised 5-year RSR (%)
Area Year
Total Male Female Total Male Female
China [46] 2003-2005 - - - 10.1 10.2 10.3
2006-2008 - - - 10.1 10.0 11.0
2009-2011 - - - 9.8 9.8 10.7
2012-2015 - - - 12.1 12.2 13.1
East China Shanghai [39] 2002-2006 15.5 16.0 14.8 - - -
Zhejiang Total [41] 2005-2010 19.1 19.5 18.0 - - -
Haining and 2003-2006 10.3 9.8 11.4 10.2 - -
Jiashan [48] 2007-2010 8.9 9.5 7.9 9.0 - -
2011-2014 10.6 11.3 8.9 10.2 - -
Jiangsu Huaian [51] 2010 8.4 8.9 6.9 - - -
Jintan District of 2012-2013 11.6 - - - - -
Changzhou [53]
Qidong [40,52] 1972-2011 4.7 4.5 5.4 - - -
1973-1977 2.8 - - - - -
1978-1982 1.4 - - - - -
1983-1987 2.6 - - - - -
1988-1992 4.7 - - - - -
1993-1997 4.7 - - - - -
1998-2002 5.1 - - - - -
2001-2007 10.0 9.8 10.6 - - -
2003-2007 7.1 - - - - -
Taiwan [47] 2004-2008 28.9 28.1 31.7 27.6 27.0 31.5
North China Beijing [45] 1982-1983 - 2.2 2.4 - - -
1987-1988 - 3.4 5.3 - - -
Hebei [49,50] Cixian 2000-2002 4.2 - - - - -
2003-2013 7.6 7.1 8.7 - - -
South China Hong Kong [28] 1996-2001 - - - 22.4 - -
Northeast China Liaoning [28] 2000-2002 - - - 10.7 8.8 15.2
-: No reports or non-available in the original articles; RSR: relative survival rate
DISCUSSION
Survival data based on clinical trials, hospital-based follow-up studies, and population-based cancer
registration are disparate in their aims, methods of survival estimation, and application. This study
collected overall or age-standardized RSRs of liver cancer worldwide so that we can describe the prognosis
of liver cancer in the general population, and make comparisons between different countries and regions.
All publications in the study were from the cancer registries or population-based survival analysis, which
aimed to provide valuable information for epidemiologists, basic scientists, oncologists, and clinical
physicians in liver cancer research.
The aim of clinical trials and hospital-based follow-up studies are quite different from that of population-
based survival studies. The survival obtained from clinical trials or studies comes from the evaluation
of certain therapeutics, and generally adopts overall survival (defined as the date from randomization
to death from any cause) and progression-free survival (defined as the date from randomization until
progression or death from any cause) as endpoints. For instance, a randomized, phase 3 clinical trial
published in the New England Journal of Medicine evaluated cabozantinib as compared with placebo in
previously treated patients with advanced hepatocellular carcinoma, and demonstrated that cabozantinib
treatment significantly prolonged survival in patients with longer overall survival and progression-free
survival (median overall survival and median progression-free survival were 10.2 months and 5.2 months,
[54]
respectively) compared to placebo (8.0 months and 1.9 months, respectively) . Hospital-based survival
or follow-up studies rely on hospital-based cancer follow-up or registries that collect survival information