Page 88 - Read Online
P. 88
Kühn et al. Energy Mater 2023;3:300020 https://dx.doi.org/10.20517/energymater.2023.07 Page 9 of 14
-2
-2
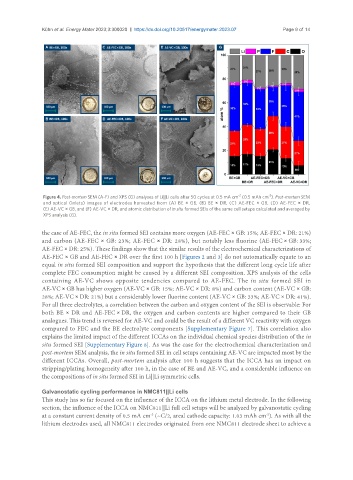
Figure 4. Post-mortem SEM (A-F) and XPS (G) analyses of Li||Li cells after 50 cycles at 0.5 mA cm (0.5 mAh cm ). Post-mortem SEM
and optical (inlets) images of electrodes harvested from (A) BE × GB, (B) BE × DR, (C) AE-FEC × GB, (D) AE-FEC × DR,
(E) AE-VC × GB, and (F) AE-VC × DR, and atomic distribution of in situ formed SEIs of the same cell setups calculated and averaged by
XPS analysis (G).
the case of AE-FEC, the in situ formed SEI contains more oxygen (AE-FEC × GB: 15%; AE-FEC × DR: 21%)
and carbon (AE-FEC × GB: 23%; AE-FEC × DR: 28%), but notably less fluorine (AE-FEC × GB: 33%;
AE-FEC × DR: 25%). These findings show that the similar results of the electrochemical characterizations of
AE-FEC × GB and AE-FEC × DR over the first 100 h [Figures 2 and 3] do not automatically equate to an
equal in situ formed SEI composition and support the hypothesis that the different long cycle life after
complete FEC consumption might be caused by a different SEI composition. XPS analysis of the cells
containing AE-VC shows opposite tendencies compared to AE-FEC. The in situ formed SEI in
AE-VC × GB has higher oxygen (AE-VC × GB: 15%; AE-VC × DR: 8%) and carbon content (AE-VC × GB:
28%; AE-VC × DR: 21%) but a considerably lower fluorine content (AE-VC × GB: 33%; AE-VC × DR: 41%).
For all three electrolytes, a correlation between the carbon and oxygen content of the SEI is observable: For
both BE × DR and AE-FEC × DR, the oxygen and carbon contents are higher compared to their GB
analogues. This trend is reversed for AE-VC and could be the result of a different VC reactivity with oxygen
compared to FEC and the BE electrolyte components [Supplementary Figure 7]. This correlation also
explains the limited impact of the different ICCAs on the individual chemical species distribution of the in
situ formed SEI [Supplementary Figure 8]. As was the case for the electrochemical characterization and
post-mortem SEM analysis, the in situ formed SEI in cell setups containing AE-VC are impacted most by the
different ICCAs. Overall, post-mortem analysis after 100 h suggests that the ICCA has an impact on
stripping/plating homogeneity after 100 h, in the case of BE and AE-VC, and a considerable influence on
the compositions of in situ formed SEI in Li||Li symmetric cells.
Galvanostatic cycling performance in NMC811||Li cells
This study has so far focused on the influence of the ICCA on the lithium metal electrode. In the following
section, the influence of the ICCA on NMC811||Li full cell setups will be analyzed by galvanostatic cycling
at a constant current density of 0.5 mA cm (~C/2, areal cathode capacity: 1.03 mAh cm ). As with all the
-2
-2
lithium electrodes used, all NMC811 electrodes originated from one NMC811 electrode sheet to achieve a