Page 72 - Read Online
P. 72
Mazzapioda et al. Energy Mater 2023;3:300019 https://dx.doi.org/10.20517/energymater.2023.03 Page 23 of 30
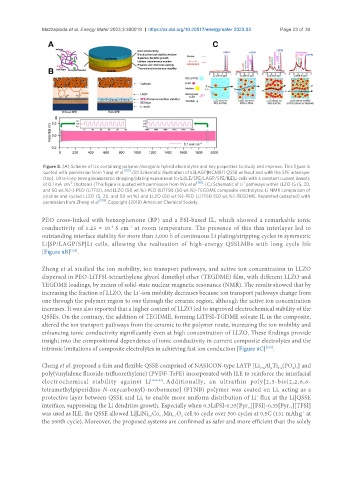
Figure 8. (A) Scheme of ILs containing polymer/inorganic hybrid electrolytes and key properties to study and improve. This figure is
quoted with permission from Yang et al. [155] . (B) Schematic illustration of Li|LAGP|NCM811 QSSB without and with the SPE interlayer
(top), Ultra-long-term galvanostatic stripping/plating experiment for Li|ILE/SPE/LAGP/SPE/ILE|Li cells with a constant current density
-2
+
of 0.1 mA cm . (bottom) (This figure is quoted with permission from Wu et al [158] . (C) Schematic of Li pathways within LLZO (5 (5, 20,
and 50 wt.%)-)-PEO (LiTFSI), and LLZO (50 wt.%)-PEO (LiTFSI) (50 wt.%)-TEGDME composite electrolytes. Li NMR comparison of
pristine and cycled LLZO (5, 20, and 50 wt.%) and LLZO (50 wt.%)-PEO (LiTFSI) (50 wt.%)-TEGDME. Reprinted (adapted) with
permission from Zheng et al. [159] . Copyright (2018) American Chemical Society.
PEO cross-linked with benzophenone (BP) and a FSI-based IL, which showed a remarkable ionic
conductivity of 1.25 × 10 S cm at room temperature. The presence of this thin interlayer led to
-3
- 1
outstanding interface stability for more than 2,000 h of continuous Li plating/stripping cycles in symmetric
Li|SP/LAGP/SP|Li cells, allowing the realisation of high-energy QSSLMBs with long cycle life
[Figure 8B] .
[158]
Zheng et al. studied the ion mobility, ion transport pathways, and active ion concentration in LLZO
dispersed in PEO-LiTFSI-tetraethylene glycol dimethyl ether (TEGDME) film, with different LLZO and
TEGDME loadings, by means of solid-state nuclear magnetic resonance (NMR). The results showed that by
increasing the fraction of LLZO, the Li -ion mobility decreases because ion transport pathways change from
+
one through the polymer region to one through the ceramic region, although the active ion concentration
increases. It was also reported that a higher content of LLZO led to improved electrochemical stability of the
QSSEs. On the contrary, the addition of TEGDME, forming LiTFSI-TGDME solvate IL in the composite,
altered the ion transport pathways from the ceramic to the polymer route, increasing the ion mobility and
enhancing ionic conductivity significantly even at high concentration of LLZO. These findings provide
insight into the compositional dependence of ionic conductivity in current composite electrolytes and the
intrinsic limitations of composite electrolytes in achieving fast ion conduction [Figure 8C] .
[159]
Cheng et al. proposed a thin and flexible QSSE comprised of NASICON-type LATP [Li Al Ti (PO ) ] and
x
2-x
1+x
4 3
poly(vinylidene fluoride-trifluorethylene) (PVDF-TrFE) incorporated with ILE to reinforce the interfacial
electrochemical stability against Li [160,161] . Additionally, an ultrathin poly[2,3-bis(2,2,6,6-
tetramethylpiperidine-N-oxycarbonyl)-norbornene] (PTNB) polymer was coated on Li, acting as a
protective layer between QSSE and Li, to enable more uniform distribution of Li flux at the Li|QSSE
+
interface, suppressing the Li dendrites growth. Especially when 0.3LiFSI-0.35[Pyr ][FSI]-0.35[Pyr ][TFSI]
14
14
was used as ILE, the QSSE allowed Li|LiNi Co Mn O cell to cycle over 500 cycles at 0.5C (131 mAh g at
-1
0.1
0.8
2
0.1
the 500th cycle). Moreover, the proposed systems are confirmed as safer and more efficient than the solely