Page 67 - Read Online
P. 67
Page 18 of 30 Mazzapioda et al. Energy Mater 2023;3:300019 https://dx.doi.org/10.20517/energymater.2023.03
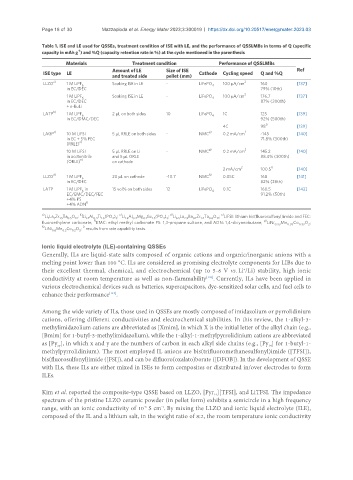
Table 1. ISE and LE used for QSSEs, treatment condition of ISE with LE, and the performance of QSSLMBs in terms of Q (specific
-1
capacity in mAh g ) and %Q (capacity retention rate in %) at the cycle mentioned in the parenthesis
Materials Treatment condition Performance of QSSLMBs
Amount of LE Size of ISE Ref
ISE type LE Cathode Cycling speed Q and %Q
and treated side pellet (mm)
a) 2
LLZO 1 M LiPF Soaking ISE in LE - LiFePO 100 μA/cm 160 [137]
6 4
in EC/DEC 79% (10th)
2
1 M LiPF Soaking ISE in LE - LiFePO 100 μA/cm 176.7 [137]
6 4
in EC/DEC 87% (200th)
+ n-BuLi
b)
LATP 1 M LiPF 2 µL on both sides 10 LiFePO 4 1C 125 [139]
6
in EC/DMC/DEC 92% (500th)
i)
4C 98 [139]
LAGP c) 10 M LiFSI 5 μL RRLE on both sides - NMC g) 0.2 mA/cm 2 ~145 [140]
in EC + 5% FEC 71.8% (300th)
(RRLE) e)
g) 2
10 M LiFSI 5 μL RRLE on Li - NMC 0.2 mA/cm 145.2 [140]
in acetonitrile and 5 μL ORLE 88.4% (300th)
e)
(ORLE) on cathode
2 i)
2 mA/cm 100.5 [140]
LLZO d) 1 M LiPF 20 µL on cathode ~10.7 NMC h) 0.05C 168 [141]
6
in EC/DEC 82% (28th)
LATP 1 M LiPF in 15 vol% on both sides 12 LiFePO 4 0.1C 160.5 [142]
6
EC/EMC/DEC/FEC 91.2% (50th)
+4% PS
f)
+4% ADN
a) b) c) d) e)
Li La Zr Ta O ; Li Al Ti (PO ) ; Li Al Mg Ge (PO ) ; Li La Ba Zr Ta O ; LiFSI: lithium bis(fluorosulfonyl)imide and FEC:
4 3
0.4
1.6
1.4
0.4
1.6
0.1
1.4
0.1
12
0.6
4 3
1.5
2.9
6.5
0.5
12
3
7
1.5
f)
g)
fluoroethylene carbonate; EMC: ethyl methyl carbonate PS: 1,3-propane sultone, and ADN: 1,4-dicyanobutane; LiNi 0.33 Mn 0.33 Co 0.33 O ;
2
h) i)
LiNi Mn Co O ; results from rate capability tests.
0.6 0.2 0.2 2
Ionic liquid electrolyte (ILE)-containing QSSEs
Generally, ILs are liquid-state salts composed of organic cations and organic/inorganic anions with a
melting point lower than 100 °C. ILs are considered as promising electrolyte components for LIBs due to
+
their excellent thermal, chemical, and electrochemical (up to 5-6 V vs. Li /Li) stability, high ionic
conductivity at room temperature as well as non-flammability . Currently, ILs have been applied in
[143]
various electrochemical devices such as batteries, supercapacitors, dye-sensitized solar cells, and fuel cells to
enhance their performance .
[144]
Among the wide variety of ILs, those used in QSSEs are mostly composed of imidazolium or pyrrolidinium
cations, offering different conductivities and electrochemical stabilities. In this review, the 1-alkyl-3-
methylimidazolium cations are abbreviated as [Xmim], in which X is the initial letter of the alkyl chain (e.g.,
[Bmim] for 1-butyl-3-methylimidazolium), while the 1-alkyl-1-methylpyrrolidinium cations are abbreviated
as [Py ], in which x and y are the numbers of carbon in each alkyl side chains (e.g., [Py ] for 1-butyl-1-
xy
14
methylpyrrolidinium). The most employed IL anions are bis(trifluoromethanesulfonyl)imide ([TFSI]),
bis(fluorosulfonyl)imide ([FSI]), and can be difluoro(oxalato)borate ([DFOB]). In the development of QSSE
with ILs, these ILs are either mixed in ISEs to form composites or distributed in/over electrodes to form
ILEs.
Kim et al. reported the composite-type QSSE based on LLZO, [Pyr ][TFSI], and LiTFSI. The impedance
14
spectrum of the pristine LLZO ceramic powder (in pellet form) exhibits a semicircle in a high frequency
-1
range, with an ionic conductivity of 10 S cm . By mixing the LLZO and ionic liquid electrolyte (ILE),
-6
composed of the IL and a lithium salt, in the weight ratio of 8:2, the room temperature ionic conductivity