Page 39 - Read Online
P. 39
Miao et al. Energy Mater 2023;3:300014 https://dx.doi.org/10.20517/energymater.2022.89 Page 21 of 31
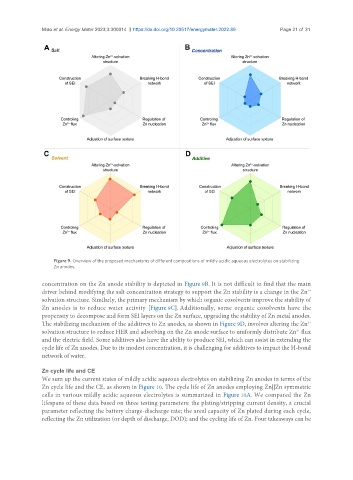
Figure 9. Overview of the proposed mechanisms of different compositions of mildly acidic aqueous electrolytes on stabilizing
Zn anodes.
concentration on the Zn anode stability is depicted in Figure 9B. It is not difficult to find that the main
2+
driver behind modifying the salt concentration strategy to support the Zn stability is a change in the Zn
solvation structure. Similarly, the primary mechanism by which organic cosolvents improve the stability of
Zn anodes is to reduce water activity [Figure 9C]. Additionally, some organic cosolvents have the
propensity to decompose and form SEI layers on the Zn surface, upgrading the stability of Zn metal anodes.
The stabilizing mechanism of the additives to Zn anodes, as shown in Figure 9D, involves altering the Zn
2+
2+
solvation structure to reduce HER and adsorbing on the Zn anode surface to uniformly distribute Zn flux
and the electric field. Some additives also have the ability to produce SEI, which can assist in extending the
cycle life of Zn anodes. Due to its modest concentration, it is challenging for additives to impact the H-bond
network of water.
Zn cycle life and CE
We sum up the current states of mildly acidic aqueous electrolytes on stabilizing Zn anodes in terms of the
Zn cycle life and the CE, as shown in Figure 10. The cycle life of Zn anodes employing Zn||Zn symmetric
cells in various mildly acidic aqueous electrolytes is summarized in Figure 10A. We compared the Zn
lifespans of these data based on three testing parameters: the plating/stripping current density, a crucial
parameter reflecting the battery charge-discharge rate; the areal capacity of Zn plated during each cycle,
reflecting the Zn utilization (or depth of discharge, DOD); and the cycling life of Zn. Four takeaways can be