Page 130 - Read Online
P. 130
Xu et al. Chem Synth 2023;3:17 https://dx.doi.org/10.20517/cs.2022.35 Page 3 of 10
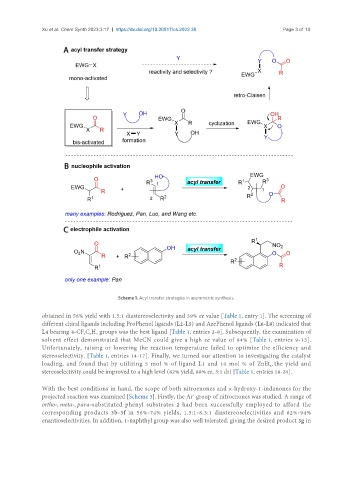
Scheme 1. Acyl transfer strategies in asymmetric synthesis.
obtained in 56% yield with 1.5:1 diastereoselectivity and 39% ee value [Table 1, entry 1]. The screening of
different chiral ligands including ProPhenol ligands (L2-L5) and AzePhenol ligands (L6-L8) indicated that
L4 bearing 4-CF C H groups was the best ligand [Table 1, entries 2-8]. Subsequently, the examination of
6
3
4
solvent effect demonstrated that MeCN could give a high ee value of 84% [Table 1, entries 9-13].
Unfortunately, raising or lowering the reaction temperature failed to optimize the efficiency and
stereoselectivity. [Table 1, entries 14-17]. Finally, we turned our attention to investigating the catalyst
loading, and found that by utilizing 5 mol % of ligand L1 and 10 mol % of ZnEt , the yield and
2
stereoselectivity could be improved to a high level (82% yield, 88% ee, 3:1 dr) [Table 1, entries 18-20].
With the best conditions in hand, the scope of both nitroenones and α-hydroxy-1-indanones for the
projected reaction was examined [Scheme 3]. Firstly, the Ar group of nitroenones was studied. A range of
1
ortho-, meta-, para-substituted phenyl substrates 2 had been successfully employed to afford the
corresponding products 3b-3f in 56%-74% yields, 1.5:1-8.3:1 diastereoselectivities and 82%-94%
enantioselectivities. In addition, 1-naphthyl group was also well tolerated, giving the desired product 3g in