Page 16 - Read Online
P. 16
Cheng et al. Cancer Drug Resist. 2025;8:46 Page 9 of 28
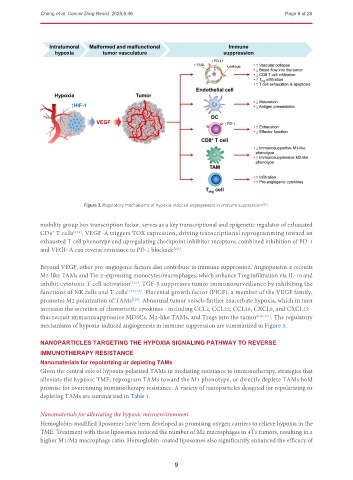
Figure 3. Regulatory mechanisms of hypoxia-induced angiogenesis in immune suppression [122] .
mobility group box transcription factor, serves as a key transcriptional and epigenetic regulator of exhausted
CD8 T cells [124] . VEGF-A triggers TOX expression, driving transcriptional reprogramming toward an
+
exhausted T cell phenotype and upregulating checkpoint inhibitor receptors; combined inhibition of PD-1
and VEGF-A can reverse resistance to PD-1 blockade .
[125]
Beyond VEGF, other pro-angiogenic factors also contribute to immune suppression. Angiopoietin-2 recruits
M2-like TAMs and Tie-2-expressing monocytes/macrophages, which enhance Treg infiltration via IL-10 and
inhibit cytotoxic T cell activation [126] . TGF-β suppresses tumor immunosurveillance by inhibiting the
functions of NK cells and T cells [127,128] . Placental growth factor (PlGF), a member of the VEGF family,
promotes M2 polarization of TAMs [129] . Abnormal tumor vessels further exacerbate hypoxia, which in turn
increases the secretion of chemotactic cytokines - including CCL2, CCL22, CCL28, CXCL8, and CXCL12 -
that recruit immunosuppressive MDSCs, M2-like TAMs, and Tregs into the tumor [130,131] . The regulatory
mechanisms of hypoxia-induced angiogenesis in immune suppression are summarized in Figure 3.
NANOPARTICLES TARGETING THE HYPOXIA SIGNALING PATHWAY TO REVERSE
IMMUNOTHERAPY RESISTANCE
Nanomaterials for repolarizing or depleting TAMs
Given the central role of hypoxia-polarized TAMs in mediating resistance to immunotherapy, strategies that
alleviate the hypoxic TME, reprogram TAMs toward the M1 phenotype, or directly deplete TAMs hold
promise for overcoming immunotherapy resistance. A variety of nanoparticles designed for repolarizing or
depleting TAMs are summarized in Table 1.
Nanomaterials for alleviating the hypoxic microenvironment
Hemoglobin-modified liposomes have been developed as promising oxygen carriers to relieve hypoxia in the
TME. Treatment with these liposomes reduced the number of M2 macrophages in 4T1 tumors, resulting in a
higher M1/M2 macrophage ratio. Hemoglobin-coated liposomes also significantly enhanced the efficacy of
9