Page 14 - Read Online
P. 14
Cheng et al. Cancer Drug Resist. 2025;8:46 Page 7 of 28
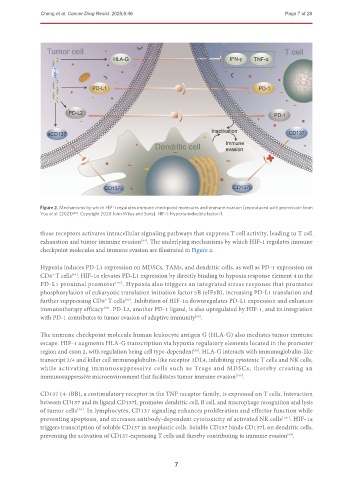
Figure 2. Mechanisms by which HIF-1 regulates immune checkpoint molecules and immune evasion [reproduced with permission from
You et al. (2021) [98] . Copyright 2020 John Wiley and Sons]. HIF-1: Hypoxia-inducible factor-1.
these receptors activates intracellular signaling pathways that suppress T cell activity, leading to T cell
exhaustion and tumor immune evasion . The underlying mechanisms by which HIF-1 regulates immune
[94]
checkpoint molecules and immune evasion are illustrated in Figure 2.
Hypoxia induces PD-L1 expression on MDSCs, TAMs, and dendritic cells, as well as PD-1 expression on
CD8 T cells . HIF-1α elevates PD-L1 expression by directly binding to hypoxia response element 4 in the
+
[94]
PD-L1 proximal promoter [95] . Hypoxia also triggers an integrated stress response that promotes
phosphorylation of eukaryotic translation initiation factor 5B (eIF5B), increasing PD-L1 translation and
further suppressing CD8 T cells . Inhibition of HIF-1α downregulates PD-L1 expression and enhances
[96]
+
immunotherapy efficacy . PD-L2, another PD-1 ligand, is also upregulated by HIF-1, and its integration
[97]
with PD-1 contributes to tumor evasion of adaptive immunity .
[98]
The immune checkpoint molecule human leukocyte antigen G (HLA-G) also mediates tumor immune
escape. HIF-1 augments HLA-G transcription via hypoxia regulatory elements located in the promoter
region and exon 2, with regulation being cell type-dependent . HLA-G interacts with immunoglobulin-like
[99]
transcript 2/4 and killer cell immunoglobulin-like receptor 2DL4, inhibiting cytotoxic T cells and NK cells,
while activating immunosuppressive cells such as Tregs and MDSCs, thereby creating an
immunosuppressive microenvironment that facilitates tumor immune evasion .
[100]
CD137 (4-1BB), a costimulatory receptor in the TNF receptor family, is expressed on T cells. Interaction
between CD137 and its ligand CD137L promotes dendritic cell, B cell, and macrophage recognition and lysis
of tumor cells . In lymphocytes, CD137 signaling enhances proliferation and effector function while
[98]
preventing apoptosis, and increases antibody-dependent cytotoxicity of activated NK cells [101] . HIF-1α
triggers transcription of soluble CD137 in neoplastic cells. Soluble CD137 binds CD137L on dendritic cells,
preventing the activation of CD137-expressing T cells and thereby contributing to immune evasion .
[98]
7