Page 75 - Read Online
P. 75
Page 10 of 27 Liu et al. Microstructures 2023;3:2023020 https://dx.doi.org/10.20517/microstructures.2023.02
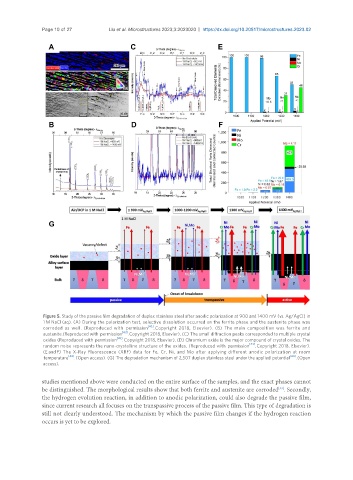
Figure 5. Study of the passive film degradation of duplex stainless steel after anodic polarization at 900 and 1400 mV (vs. Ag/AgCl) in
1 M NaCl (aq). (A) During the polarization test, selective dissolution occurred on the ferrite phase and the austenite phase was
corroded as well. (Reproduced with permission [43] . Copyright 2018, Elsevier). (B) The main composition was ferrite and
austenite.(Reproduced with permission [43] . Copyright 2018, Elsevier). (C) The small diffraction peaks corresponded to multiple crystal
oxides (Reproduced with permission [43] . Copyright 2018, Elsevier). (D) Chromium oxide is the major compound of crystal oxides. The
random noise represents the nano-crystalline structure of the oxides. (Reproduced with permission [43] . Copyright 2018, Elsevier).
(E and F) The X-Ray Fluorescence (XRF) data for Fe, Cr, Ni, and Mo after applying different anodic polarization at room
temperature [44] . (Open access). (G) The degradation mechanism of 2,507 duplex stainless steel under the applied potential [44] . (Open
access).
studies mentioned above were conducted on the entire surface of the samples, and the exact phases cannot
[43]
be distinguished. The morphological results show that both ferrite and austenite are corroded . Secondly,
the hydrogen evolution reaction, in addition to anodic polarization, could also degrade the passive film,
since current research all focuses on the transpassive process of the passive film. This type of degradation is
still not clearly understood. The mechanism by which the passive film changes if the hydrogen reaction
occurs is yet to be explored.