Page 256 - Read Online
P. 256
Page 8 of 19 Mondal et al. Neuroimmunol Neuroinflammation 2018;5:34 I http://dx.doi.org/10.20517/2347-8659.2018.13
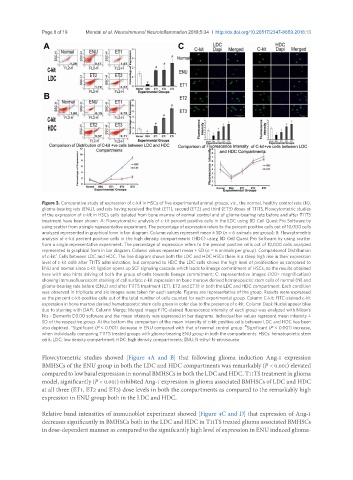
Figure 3. Comparative study of expression of c-kit in HSCs of five experimental animal groups, viz., the normal, healthy control rats (N),
glioma-bearing rats (ENU), and rats having received the first (ET1), second (ET2) and third (ET3) doses of T11TS. Flowcytometric studies
of the expression of c-kit in HSCs cells isolated from bone marrow of normal control and of glioma-bearing rats before and after T11TS
treatment have been shown. A: Flowcytometric analysis of c-kit percent positive cells in the LDC using BD Cell Quest Pro Software by
using scatter from a single representative experiment. The percentage of expression refers to the percent positive cells out of 10,000 cells
analyzed represented in graphical form in bar diagram. Column values represent mean ± SD (n = 6 animals per group); B: Flowcytometric
analysis of c-kit percent positive cells in the high density compartments (HDC) using BD Cell Quest Pro Software by using scatter
from a single representative experiment. The percentage of expression refers to the percent positive cells out of 10,000 cells analyzed
represented in graphical form in bar diagram. Column values represent mean ± SD (n = 6 animals per group). Comparisonof Distribution
+
of c-kit Cells between LDC and HDC. The line diagram shows both the LDC and HDC HSCs there is a steep high rise in their expression
level of c-kit cells after T11TS administration, but compared to HDC the LDC cells shows the high level of proliferation as compared to
ENU and normal since c-kit ligation opens up SCF signaling cascade which leads to lineage commitment of HSCs, so the results obtained
here with also hints driving of both the group of cells towards lineage commitment; C: representative images (100× magnification)
showing immunofluorescent staining of cell surface c-kit expression on bone marrow derived hematopoietic stem cells of normal (N) and
glioma-bearing rats before (ENU) and after T11TS treatment (ET1, ET2 and ET3) in both the LDC and HDC compartment. Each condition
was observed in triplicate and six images were taken for each sample. Figures are representative of the group. Results were expressed
as the percent c-kit-positive cells out of the total number of cells counted for each experimental group. Column C-kit: FITC-stained c-kit
expression in bone marrow derived hematopoietic stem cells green in color due to the presence of c-kit. Column Dapi: Nuclei appear blue
due to staining with DAPI. Column Merge: Merged image FITC-stained fluorescence intensity of each group was analyzed with Nikon’s
Nis - Elements D3.00 software and the mean intensity was expressed in bar diagrams. Individual bar values represent mean intensity ±
SD of the respective group. At the bottom the comparison of the mean intensity of c-kit positive cells between LDC and HDC has been
#
also depicted. *Significant (P < 0.001) decrease in ENU compared with that of normal control group. Significant (P < 0.001) increase,
when individually comparing T11TS treated groups with glioma-bearing ENU group in both the compartments. HSCs: hematopoietic stem
cells; LDC: low density compartment; HDC: high density compartments; ENU: N-ethyl-N-nitrosourea
Flowcytometric studies showed [Figure 4A and B] that following glioma induction Ang-1 expression
BMHSCs of the ENU group in both the LDC and HDC compartments was remarkably (P < 0.001) elevated
compared to low basal expression in normal BMHSCs in both the LDC and HDC. T11TS treatment in glioma
model, significantly (P < 0.001) inhibited Ang-1 expression in glioma associated BMHSCs of LDC and HDC
at all three (ET1, ET2 and ET3) dose levels in both the compartments as compared to the remarkably high
expression in ENU group both in the LDC and HDC.
Relative band intensities of immunoblot experiment showed [Figure 4C and D] that expression of Ang-1
decreases significantly in BMHSCs both in the LDC and HDC in T11TS treated glioma associated BMHSCs
in dose-dependent manner as compared to the significantly high level of expression in ENU induced glioma-