Page 701 - Read Online
P. 701
Page 4 of 10 Rodinò et al. Mini-invasive Surg 2020;4:70 I http://dx.doi.org/10.20517/2574-1225.2020.55
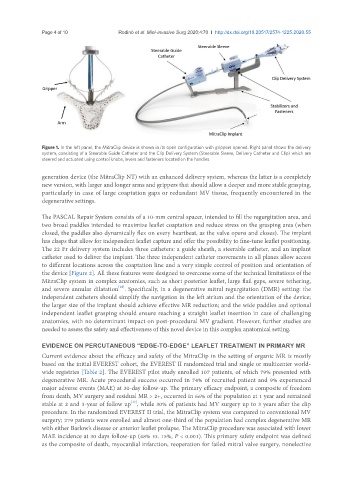
Figure 1. In the left panel, the MitraClip device is shown in its open configuration with grippers opened. Right panel shows the delivery
system, consisting of a Steerable Guide Catheter and the Clip Delivery System (Steerable Sleeve, Delivery Catheter and Clip) which are
steered and actuated using control knobs, levers and fasteners located on the handles
generation device (the MitraClip NT) with an enhanced delivery system, whereas the latter is a completely
new version, with larger and longer arms and grippers that should allow a deeper and more stable grasping,
particularly in case of large coaptation gaps or redundant MV tissue, frequently encountered in the
degenerative settings.
The PASCAL Repair System consists of a 10-mm central spacer, intended to fill the regurgitation area, and
two broad paddles intended to maximize leaflet coaptation and reduce stress on the grasping area (when
closed, the paddles also dynamically flex on every heartbeat, as the valve opens and closes). The implant
has clasps that allow for independent leaflet capture and offer the possibility to fine-tune leaflet positioning.
The 22 Fr delivery system includes three catheters: a guide sheath, a steerable catheter, and an implant
catheter used to deliver the implant. The three independent catheter movements in all planes allow access
to different locations across the coaptation line and a very simple control of position and orientation of
the device [Figure 2]. All these features were designed to overcome some of the technical limitations of the
MitraClip system in complex anatomies, such as short posterior leaflet, large flail gaps, severe tethering,
[16]
and severe annular dilatation . Specifically, in a degenerative mitral regurgitation (DMR) setting: the
independent catheters should simplify the navigation in the left atrium and the orientation of the device;
the larger size of the implant should achieve effective MR reduction; and the wide paddles and optional
independent leaflet grasping should ensure reaching a straight leaflet insertion in case of challenging
anatomies, with no determinant impact on post-procedural MV gradient. However, further studies are
needed to assess the safety and effectiveness of this novel device in this complex anatomical setting.
EVIDENCE ON PERCUTANEOUS “EDGE-TO-EDGE” LEAFLET TREATMENT IN PRIMARY MR
Current evidence about the efficacy and safety of the MitraClip in the setting of organic MR is mostly
based on the initial EVEREST cohort, the EVEREST II randomized trial and single or multicenter world-
wide registries [Table 2]. The EVEREST pilot study enrolled 107 patients, of which 79% presented with
degenerative MR. Acute procedural success occurred in 74% of recruited patient and 9% experienced
major adverse events (MAE) at 30-day follow-up. The primary efficacy endpoint, a composite of freedom
from death, MV surgery and residual MR > 2+, occurred in 66% of the population at 1 year and remained
[13]
stable at 2 and 3-year of follow up , while 30% of patients had MV surgery up to 3 years after the clip
procedure. In the randomized EVEREST II trial, the MitraClip system was compared to conventional MV
surgery; 279 patients were enrolled and almost one-third of the population had complex degenerative MR
with either Barlow’s disease or anterior leaflet prolapse. The MitraClip procedure was associated with lower
MAE incidence at 30 days follow-up (48% vs. 15%, P < 0.001). This primary safety endpoint was defined
as the composite of death, myocardial infarction, reoperation for failed mitral valve surgery, nonelective