Page 71 - Read Online
P. 71
Page 12 of 25 Meng et al. J. Mater. Inf. 2025, 5, 3 https://dx.doi.org/10.20517/jmi.2024.74
Figure 5. (A) Free energy profiles and corresponding intermediate structures on V @g-C N andNi @g-C N , Color scheme: H, white; C,
2
4
3
4
3
2
gray; N, blue; V, red; Ni, pink. Reprinted with permission from Ref. [93] . Copyright © 2022 Wiley-VCH GmbH; (B) N≡N bond lengths with
* a
and without H generated on the X site and the relationship between G and E NNH* on X/Fe-N-C (X = Pd, Ir, Pt), with an inset showing
H*
* [94]
the schematic diagram of the cleavage of X−H bond to form NNH . Reprinted with permission from Ref. . Copyright © 2023 Wiley-
VCH GmbH.
[101]
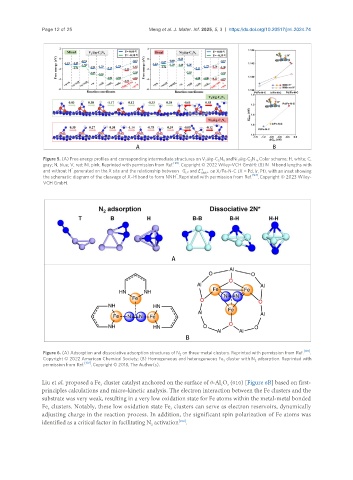
Figure 6. (A) Adsorption and dissociative adsorption structures of N on three-metal clusters. Reprinted with permission from Ref. .
2
Copyright © 2022 American Chemical Society; (B) Homogeneous and heterogeneous Fe cluster with N adsorption. Reprinted with
3 2
[102]
permission from Ref. . Copyright © 2018, The Author(s).
Liu et al. proposed a Fe cluster catalyst anchored on the surface of θ-Al O (010) [Figure 6B] based on first-
3
2
3
principles calculations and micro-kinetic analysis. The electron interaction between the Fe clusters and the
substrate was very weak, resulting in a very low oxidation state for Fe atoms within the metal-metal bonded
Fe clusters. Notably, these low oxidation state Fe clusters can serve as electron reservoirs, dynamically
3
3
adjusting charge in the reaction process. In addition, the significant spin polarization of Fe atoms was
identified as a critical factor in facilitating N activation .
[102]
2