Page 38 - Read Online
P. 38
Zhang et al. J Mater Inf 2024;4:1 https://dx.doi.org/10.20517/jmi.2023.34 Page 7 of 14
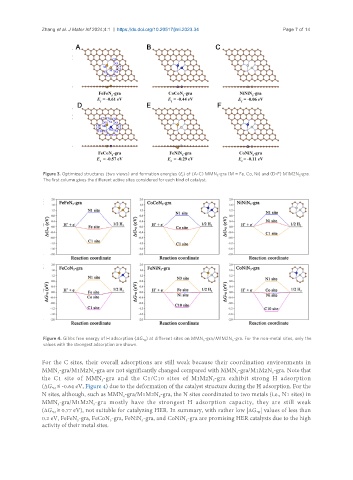
Figure 3. Optimized structures (two views) and formation energies (E ) of (A-C) MMN -gra (M = Fe, Co, Ni) and (D-F) M1M2N -gra.
f 5 5
The first column gives the different active sites considered for each kind of catalyst.
Figure 4. Gibbs free energy of H adsorption (ΔG ) at different sites on MMN -gra/M1M2N -gra. For the non-metal sites, only the
*H 5 5
values with the strongest adsorption are shown.
For the C sites, their overall adsorptions are still weak because their coordination environments in
MMN -gra/M1M2N -gra are not significantly changed compared with MMN -gra/M1M2N -gra. Note that
6
5
5
6
the C1 site of MMN -gra and the C1/C10 sites of M1M2N -gra exhibit strong H adsorption
5
5
(ΔG ≤ -0.64 eV, Figure 4) due to the deformation of the catalyst structure during the H adsorption. For the
*H
N sites, although, such as MMN -gra/M1M2N -gra, the N sites coordinated to two metals (i.e., N1 sites) in
6
6
MMN -gra/M1M2N -gra mostly have the strongest H adsorption capacity, they are still weak
5
5
(ΔG ≥ 0.77 eV), not suitable for catalyzing HER. In summary, with rather low |ΔG | values of less than
*H
*H
0.2 eV, FeFeN -gra, FeCoN -gra, FeNiN -gra, and CoNiN -gra are promising HER catalysts due to the high
5
5
5
5
activity of their metal sites.