Page 203 - Read Online
P. 203
kinases, 30% of new efforts by pharmaceutical companies with cytotoxic, hormonal or targeted agents. BMS-754807
are dedicated to develop new kinase inhibitors with many in combination with docetaxel showed more than
already approved or in clinical trials. Few examples additive effi cacy in triple negative breast cancer. There
[45]
of TKIs for GFRs are enlisted in Table 4. Chemo- or are now several clinical trials (phase I and II) ongoing
radio-therapeutic agents are often combined with TKIs with BMS-754807-including combinations. Co-targeting
to increase therapeutic effi cacy. The IGF1R inhibitor, of IGF1R and IR with OSI-906 inhibitor showed
BMS-754807 showed higher effi cacy when combined superior antitumor activity compared to targeting IGF1R
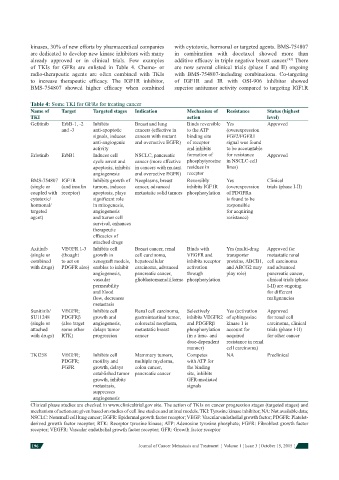
Table 4: Some TKI for GFRs for treating cancer
Name of Target Targeted stages Indication Mechanism of Resistance Status (highest
TKI action level)
Gefi tinib ErbB-1, -2 Inhibits Breast and lung Binds reversible Yes Approved
and -3 anti-apoptotic cancers (effective in to the ATP (overexpression
signals, induces cancers with mutant binding site FGF2/FGFR1
anti-angiogenic and overactive EGFR) of receptor signal was found
activity and inhibits to be accountable
Erlotinib ErbB1 Induces cell NSCLC, pancreatic formation of for resistance Approved
cycle arrest and cancer (more effective phosphytyrosine in NSCLC cell
apoptosis, inhibits in cancers with mutant residues in lines)
angiogenesis and overactive EGFR) receptor
BMS-754807 IGF1R Inhibits growth of Neoplasms, breast Reversibly Yes Clinical
(single or (and insulin tumors, induces cancer, advanced inhibits IGF1R (overexpression trials (phase I-II)
coupled with receptor) apoptosis, plays metastatic solid tumors phosphorylation of PDGFRα
cytotoxic/ signifi cant role is found to be
hormonal/ in mitogenesis, responsible
targeted angiogenesis for acquiring
agent) and tumor cell resistance)
survival, enhances
therapeutic
effi cacies of
attached drugs
Axitinib VEGFR 1-3 Inhibits cell Breast cancer, renal Binds with Yes (multi-drug Approved for
(single or (thought growth in cell carcinoma, VEGFR and transporter metastatic renal
combined to act on xenograft models, hepatocellular inhibits receptor proteins, ABCB1, cell carcinoma
with drugs) PDGFR also) enables to inhibit carcinoma, advanced activation and ABCG2 may and advanced
angiogenesis, pancreatic cancer, through play role) pancreatic cancer,
vascular glioblastomamultiforme phosphorylation clinical trials (phase
permeability I-II) are ongoing
and blood for different
fl ow, decreases malignancies
metastasis
Sunitinib/ VEGFR; Inhibits cell Renal cell carcinoma, Selectively Yes (activation Approved
SU11248 PDGFRβ growth and gastrointestinal tumor, inhibits VEGFR2 of sphingosine for renal cell
(single or (also target angiogenesis, colorectal neoplasm, and PDGFRβ kinase 1 is carcinoma, clinical
attached some other delays tumor metastatic breast phosphorylation account for trials (phase I-II)
with drugs) RTK) progression cancer (in a time- and acquired for other cancer
dose-dependent resistance in renal
manner) cell carcinoma)
TKI258 VEGFR; Inhibits cell Mammary tumors, Competes NA Preclinical
PDGFR; motility and multiple myeloma, with ATP for
FGFR growth, delays colon cancer, the binding
established tumor pancreatic cancer site, inhibits
growth, inhibits GFR-mediated
metastasis, signals
suppresses
angiogenesis
Clinical phase studies are checked in www.clinicaltrial.gov site. The action of TKIs on cancer progression stages (targeted stages) and
mechanism of action are given based on studies of cell line studies and animal models. TKI: Tyrosine kinase inhibitor; NA: Not available data;
NSCLC: Nonsmall cell lung cancer; EGFR: Epidermal growth factor receptor; VEGF: Vascular endothelial growth factor; PDGFR: Platelet-
derived growth factor receptor; RTK: Receptor tyrosine kinase; ATP: Adenosine tyrosine phosphate; FGFR: Fibroblast growth factor
receptor; VEGFR: Vascular endothelial growth factor receptor; GFR: Growth factor receptor
196 Journal of Cancer Metastasis and Treatment ¦ Volume 1 ¦ Issue 3 ¦ October 15, 2015 ¦