Page 220 - Read Online
P. 220
Berge et al. Clinical outcomes of direct-acting antivirals
Clinical decompensations publication has suggested that there is a higher risk
of tumor recurrence in patients with hepatocellular
1.0 [15]
carcinoma treated with direct-acting antivirals , while
this association was not found in another prospective
Free from clinical decompensations 0.6 may have on the development of hepatocellular
. It is important to continue investigating this
study
[16]
0.8
relationship, developing long-term follow-up studies,
in order to clarify the effect that direct-acting antivirals
carcinoma and clinical decompensations so that we
can better understand and explain to our patients what
they can expect after achieving sustained virological
0.4
response.
In our study, the development of hepatocellular
0.2
3.6%, as high as what it is expected for untreated
. Thus, there seems to be no
cirrhotic patients
[4,5]
0.0 carcinoma in a median of 16 months of follow-up is
benefits in the short term in this aspect. Undoubtedly,
0 5 10 15 20 25 the study has many limitations as it is retrospective, it
Follow-up time (months) was performed in a single center, and it has a relatively
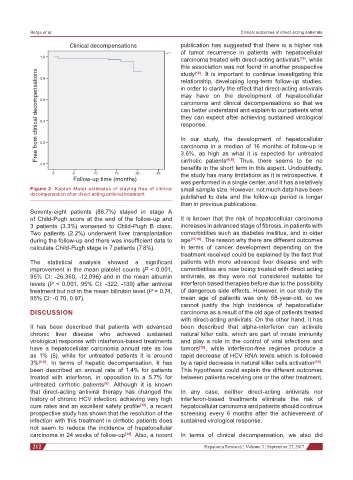
Figure 2: Kaplan Meier estimates of staying free of clinical small sample size. However, not much data have been
decompensation after direct-acting antiviral treatment
published to date and the follow-up period is longer
than in previous publications.
Seventy-eight patients (86.7%) stayed in stage A
of Child-Pugh score at the end of the follow-up and It is known that the risk of hepatocellular carcinoma
3 patients (3.3%) worsened to Child-Pugh B class. increases in advanced stage of fibrosis, in patients with
Two patients (2.2%) underwent liver transplantation comorbidities such as diabetes mellitus, and in older
during the follow-up and there was insufficient data to age [17,18] . The reason why there are different outcomes
calculate Child-Pugh stage in 7 patients (7.8%). in terms of cancer development depending on the
treatment received could be explained by the fact that
The statistical analysis showed a significant patients with more advanced liver disease and with
improvement in the mean platelet counts (P < 0.001, comorbidities are now being treated with direct acting
95% CI: -26,360, -12,096) and in the mean albumin antivirals, as they were not considered suitable for
levels (P < 0.001, 95% CI: -322, -130) after antiviral interferon based therapies before due to the possibility
treatment but not in the mean bilirubin level (P = 0.74, of dangerous side effects. However, in our study the
95% CI: -0.70, 0.97). mean age of patients was only 58-year-old, so we
cannot justify the high incidence of hepatocellular
DISCUSSION carcinoma as a result of the old age of patients treated
with direct-acting antivirals. On the other hand, it has
It has been described that patients with advanced been described that alpha-interferon can activate
chronic liver disease who achieved sustained natural killer cells, which are part of innate immunity
virological response with interferon-based treatments and play a role in the control of viral infections and
have a hepatocellular carcinoma annual rate as low tumors [19] , while interferon-free regimes produce a
as 1% (6), while for untreated patients it is around rapid decrease of HCV RNA levels which is followed
3% [5,6] . In terms of hepatic decompensation, it has by a rapid decrease in natural killer cells activation [20] .
been described an annual rate of 1.4% for patients This hypothesis could explain the different outcomes
treated with interferon, in opposition to a 5.7% for between patients receiving one or the other treatment.
untreated cirrhotic patients . Although it is known
[6]
that direct-acting antiviral therapy has changed the In any case, neither direct-acting antivirals nor
history of chronic HCV infection, achieving very high interferon-based treatments eliminate the risk of
cure rates and an excellent safety profile [12] , a recent hepatocellular carcinoma and patients should continue
prospective study has shown that the resolution of the screening every 6 months after the achievement of
infection with this treatment in cirrhotic patients does sustained virological response.
not seem to reduce the incidence of hepatocellular
carcinoma in 24 weeks of follow-up [14] . Also, a recent In terms of clinical decompensation, we also did
212 Hepatoma Research ¦ Volume 3 ¦ September 27, 2017