Page 250 - Read Online
P. 250
Page 20 of 33 Girase et al. Energy Mater. 2025, 5, 500132 https://dx.doi.org/10.20517/energymater.2025.14
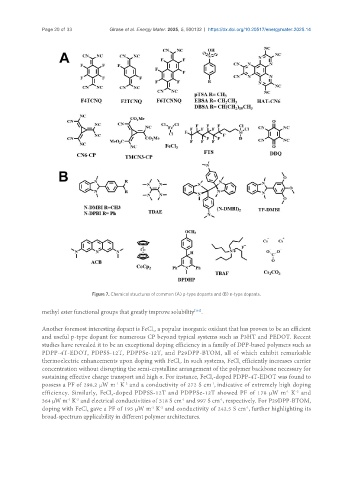
Figure 7. Chemical structures of common (A) p-type dopants and (B) n-type dopants.
methyl ester functional groups that greatly improve solubility .
[161]
Another foremost interesting dopant is FeCl , a popular inorganic oxidant that has proven to be an efficient
3
and useful p-type dopant for numerous CP beyond typical systems such as P3HT and PEDOT. Recent
studies have revealed it to be an exceptional doping efficiency in a family of DPP-based polymers such as
PDPP-4T-EDOT, PDPSS-12T, PDPPSe-12T, and P29DPP-BTOM, all of which exhibit remarkable
thermoelectric enhancements upon doping with FeCl . In such systems, FeCl efficiently increases carrier
3
3
concentration without disrupting the semi-crystalline arrangement of the polymer backbone necessary for
sustaining effective charge transport and high σ. For instance, FeCl -doped PDPP-4T-EDOT was found to
3
-1
-2
possess a PF of 298.2 μW m K and a conductivity of 272 S cm , indicative of extremely high doping
-1
efficiency. Similarly, FeCl -doped PDPSS-12T and PDPPSe-12T showed PF of 178 μW m K and
-2
-1
3
-1
-1
-1
364 μW m K and electrical conductivities of 318 S cm and 997 S cm , respectively. For P29DPP-BTOM,
-2
doping with FeCl gave a PF of 195 μW m K and conductivity of 242.5 S cm , further highlighting its
-1
-2
-1
3
broad-spectrum applicability in different polymer architectures.