Page 117 - Read Online
P. 117
Page 6 of 12 Cui et al. Energy Mater 2023;3:300034 https://dx.doi.org/10.20517/energymater.2023.19
+
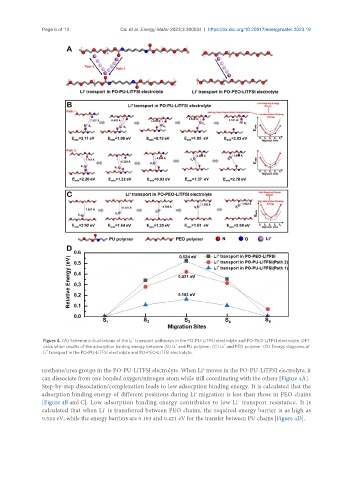
Figure 4. (A) Schematic illustrations of the Li transport pathways in the PO-PU-LiTFSI electrolyte and PO-PEO-LiTFSI electrolyte. DFT
+
+
calculation results of the adsorption binding energy between (B) Li and PU polymer, (C) Li and PEO polymer. (D) Energy diagrams of
+
Li transport in the PO-PU-LiTFSI electrolyte and PO-PEO-LiTFSI electrolyte.
urethane/urea groups in the PO-PU-LiTFSI electrolyte. When Li moves in the PO-PU-LiTFSI electrolyte, it
+
can dissociate from one bonded oxygen/nitrogen atom while still coordinating with the others [Figure 4A].
Step-by-step dissociation/complexation leads to low adsorption binding energy. It is calculated that the
adsorption binding energy of different positions during Li migration is less than those in PEO chains
+
[Figure 4B and C]. Low adsorption binding energy contributes to low Li transport resistance. It is
+
calculated that when Li is transferred between PEO chains, the required energy barrier is as high as
+
0.524 eV, while the energy barriers are 0.163 and 0.421 eV for the transfer between PU chains [Figure 4D].