Page 116 - Read Online
P. 116
Cui et al. Energy Mater 2023;3:300034 https://dx.doi.org/10.20517/energymater.2023.19 Page 5 of 12
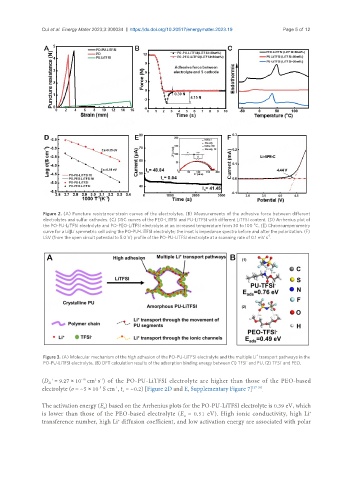
Figure 2. (A) Puncture resistance-strain curves of the electrolytes. (B) Measurements of the adhesive force between different
electrolytes and sulfur cathodes. (C) DSC curves of the PEO-LiTFSI and PU-LiTFSI with different LiTFSI content. (D) Arrhenius plot of
o
the PO-PU-LiTFSI electrolyte and PO-PEO-LiTFSI electrolyte at an increased temperature from 30 to 100 C. (E) Chronoamperometry
curve for a Li||Li symmetric cell using the PO-PU-LiTFSI electrolyte; the inset is impedance spectra before and after the polarization. (F)
-1
LSV (from the open circuit potential to 5.0 V) profile of the PO-PU-LiTFSI electrolyte at a scanning rate of 0.1 mV s .
+
Figure 3. (A) Molecular mechanism of the high adhesion of the PO-PU-LiTFSI electrolyte and the multiple Li transport pathways in the
- -
PO-PU-LiTFSI electrolyte. (B) DFT calculation results of the adsorption binding energy between (1) TFSI and PU, (2) TFSI and PEO.
(D = 9.27 × 10 cm s ) of the PO-PU-LiTFSI electrolyte are higher than those of the PEO-based
-10
+
2 -1
Li
-1
electrolyte (σ = ~5 × 10 S cm , t = ~0.2) [Figure 2D and E, Supplementary Figure 7] [27-29] .
-5
+
The activation energy (E ) based on the Arrhenius plots for the PO-PU-LiTFSI electrolyte is 0.39 eV, which
a
+
is lower than those of the PEO-based electrolyte (E = 0.51 eV). High ionic conductivity, high Li
a
+
transference number, high Li diffusion coefficient, and low activation energy are associated with polar