Page 77 - Read Online
P. 77
Choi et al. Cancer Drug Resist. 2026;9:12 Page 3 of 20
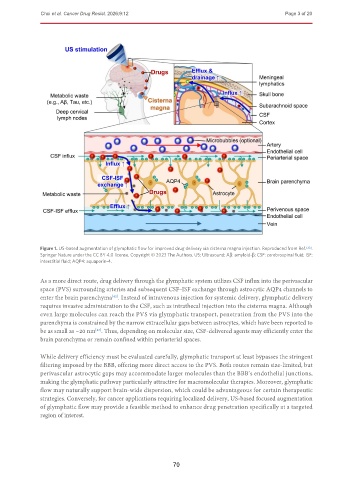
Figure 1. US-based augmentation of glymphatic flow for improved drug delivery via cisterna magna injection. Reproduced from Ref. [33] .
Springer Nature under the CC BY 4.0 license. Copyright © 2023 The Authors. US: Ultrasound; Aβ: amyloid-β; CSF: cerebrospinal fluid; ISF:
interstitial fluid; AQP4: aquaporin-4.
As a more direct route, drug delivery through the glymphatic system utilizes CSF influx into the perivascular
space (PVS) surrounding arteries and subsequent CSF–ISF exchange through astrocytic AQP4 channels to
enter the brain parenchyma . Instead of intravenous injection for systemic delivery, glymphatic delivery
[42]
requires invasive administration to the CSF, such as intrathecal injection into the cisterna magna. Although
even large molecules can reach the PVS via glymphatic transport, penetration from the PVS into the
parenchyma is constrained by the narrow extracellular gaps between astrocytes, which have been reported to
be as small as ~20 nm . Thus, depending on molecular size, CSF-delivered agents may efficiently enter the
[43]
brain parenchyma or remain confined within periarterial spaces.
While delivery efficiency must be evaluated carefully, glymphatic transport at least bypasses the stringent
filtering imposed by the BBB, offering more direct access to the PVS. Both routes remain size-limited, but
perivascular astrocytic gaps may accommodate larger molecules than the BBB’s endothelial junctions,
making the glymphatic pathway particularly attractive for macromolecular therapies. Moreover, glymphatic
flow may naturally support brain-wide dispersion, which could be advantageous for certain therapeutic
strategies. Conversely, for cancer applications requiring localized delivery, US-based focused augmentation
of glymphatic flow may provide a feasible method to enhance drug penetration specifically at a targeted
region of interest.
70