Page 136 - Read Online
P. 136
Chen et al. Microstructures 2023;3:2023025 https://dx.doi.org/10.20517/microstructures.2023.12 Page 5 of 31
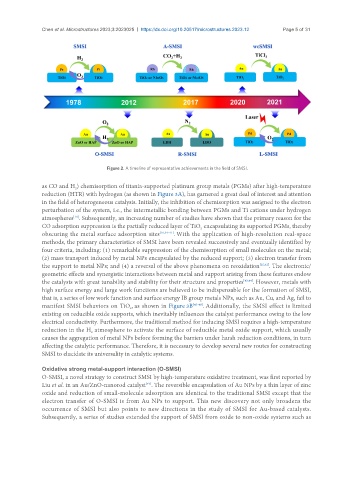
Figure 2. A timeline of representative achievements in the field of SMSI.
as CO and H ) chemisorption of titania-supported platinum group metals (PGMs) after high-temperature
2
reduction (HTR) with hydrogen (as shown in Figure 3A), has garnered a great deal of interest and attention
in the field of heterogeneous catalysis. Initially, the inhibition of chemisorption was assigned to the electron
perturbation of the system, i.e., the intermetallic bonding between PGMs and Ti cations under hydrogen
atmospheres . Subsequently, an increasing number of studies have shown that the primary reason for the
[10]
CO adsorption suppression is the partially reduced layer of TiO encapsulating its supported PGMs, thereby
2
obscuring the metal surface adsorption sites [10,39-41] . With the application of high-resolution real-space
methods, the primary characteristics of SMSI have been revealed successively and eventually identified by
four criteria, including: (1) remarkable suppression of the chemisorption of small molecules on the metal;
(2) mass transport induced by metal NPs encapsulated by the reduced support; (3) electron transfer from
the support to metal NPs; and (4) a reversal of the above phenomena on reoxidation [25,42] . The electronic/
geometric effects and synergistic interactions between metal and support arising from these features endow
the catalysts with great tunability and stability for their structure and properties [43,44] . However, metals with
high surface energy and large work functions are believed to be indispensable for the formation of SMSI,
that is, a series of low work function and surface energy IB group metals NPs, such as Au, Cu, and Ag, fail to
manifest SMSI behaviors on TiO , as shown in Figure 3B [45-48] . Additionally, the SMSI effect is limited
2
existing on reducible oxide supports, which inevitably influences the catalyst performance owing to the low
electrical conductivity. Furthermore, the traditional method for inducing SMSI requires a high-temperature
reduction in the H atmosphere to activate the surface of reducible metal oxide support, which usually
2
causes the aggregation of metal NPs before forming the barriers under harsh reduction conditions, in turn
affecting the catalytic performance. Therefore, it is necessary to develop several new routes for constructing
SMSI to elucidate its universality in catalytic systems.
Oxidative strong metal-support interaction (O-SMSI)
O-SMSI, a novel strategy to construct SMSI by high-temperature oxidative treatment, was first reported by
Liu et al. in an Au/ZnO-nanorod catalyst . The reversible encapsulation of Au NPs by a thin layer of zinc
[49]
oxide and reduction of small-molecule adsorption are identical to the traditional SMSI except that the
electron transfer of O-SMSI is from Au NPs to support. This new discovery not only broadens the
occurrence of SMSI but also points to new directions in the study of SMSI for Au-based catalysts.
Subsequently, a series of studies extended the support of SMSI from oxide to non-oxide systems such as