Page 63 - Read Online
P. 63
Page 4 of 17 Xiao et al. Microstructures 2023;3:2023006 https://dx.doi.org/10.20517/microstructures.2022.26
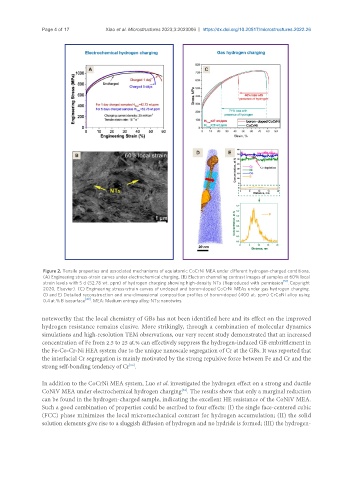
Figure 2. Tensile properties and associated mechanisms of equiatomic CoCrNi MEA under different hydrogen-charged conditions.
(A) Engineering stress-strain curves under electrochemical charging. (B) Electron channeling contrast images of samples at 60% local
strain levels with 5 d (52.78 wt. ppm) of hydrogen charging showing high-density NTs (Reproduced with permission [51] . Copyright
2020, Elsevier). (C) Engineering stress-strain curves of undoped and boron-doped CoCrNi MEAs under gas hydrogen charging.
(D and E) Detailed reconstruction and one-dimensional composition profiles of boron-doped (400 at. ppm) CrCoNi alloy using
0.4 at.% B isosurface [49] . MEA: Medium entropy alloy; NTs: nanotwins.
noteworthy that the local chemistry of GBs has not been identified here and its effect on the improved
hydrogen resistance remains elusive. More strikingly, through a combination of molecular dynamics
simulations and high-resolution TEM observations, our very recent study demonstrated that an increased
concentration of Fe from 2.5 to 25 at.% can effectively suppress the hydrogen-induced GB embrittlement in
the Fe-Co-Cr-Ni HEA system due to the unique nanoscale segregation of Cr at the GBs. It was reported that
the interfacial Cr segregation is mainly motivated by the strong repulsive force between Fe and Cr and the
strong self-bonding tendency of Cr .
[56]
In addition to the CoCrNi MEA system, Luo et al. investigated the hydrogen effect on a strong and ductile
CoNiV MEA under electrochemical hydrogen charging . The results show that only a marginal reduction
[52]
can be found in the hydrogen-charged sample, indicating the excellent HE resistance of the CoNiV MEA.
Such a good combination of properties could be ascribed to four effects: (I) the single face-centered cubic
(FCC) phase minimizes the local micromechanical contrast for hydrogen accumulation; (II) the solid
solution elements give rise to a sluggish diffusion of hydrogen and no hydride is formed; (III) the hydrogen-