Page 170 - Read Online
P. 170
Dastidar et al. Vessel Plus 2020;4:14 I http://dx.doi.org/10.20517/2574-1209.2019.36 Page 17 of 29
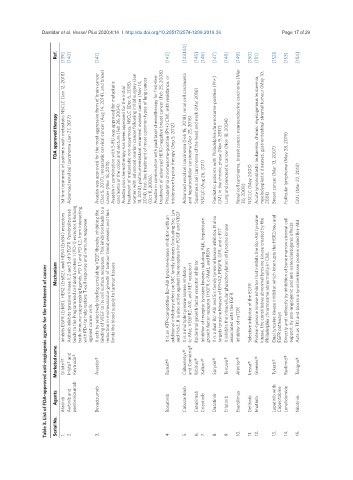
Ref. [139] [140] [141] [142] [143,144] [145] [146] [147] [148] [149] [150] [151] [152] [153] [154]
FDA approved therapy 1st-line treatment of patients with metastatic NSCLC (Jan 12, 2018) Advanced renal cell carcinoma (Jan 27, 2017) Avastin was approved for the most aggressive form of brain cancer (Dec 5, 2017), metastatic cervical cancer (Aug 14, 2014), and breast cancer (Nov 18, 2011). Avastin in combination with 5-FU was approved for metastatic carcinoma of the colon and rectum (Feb 26, 2004). Avastin plus chemotherapy has been approved for
Mechanism Inhibits EGFR (ErbB1), HER2 (ErbB2), and HER4 (ErbB4) receptors Axitinib inhibits tyrosine kinase 1, 2 and 3 of VEGFR. Pembrolizumab binds to the Programmed cell death protein 1 (PD-1) receptor, blocking both immune-suppressing ligands, PD-L1 and PD-L2, from interacting with PD-1 to help restore T-cell response and immune response It acts by selectively binding circulating VEGF, thereby inhibiting the binding of VEGF to its cell surface
Table 3. List of FDA-approved anti-angiogenic agents for the treatment of cancer
against cancer cells limits the blood supply to tumour tissues It is a multiple tyrosine kinase inhibitor (c-Met, VEGFR2, AXL and RET receptor) Epidermal growth factor receptor inhibitor associated with the EGFR Inhibitor of mTOR Selective inhibitor of the EGFR EGFR pathways
Marketed name Gilotrif® Inlyta® and Keytruda® Avastin® Busulif® Cabometyx® and Cometriq® Erbitux® Xalkori® Sprycel® Tercava® Afinitor® Iressa® Gleevec® Tykerb® Revlimid® Tasigna®
Agents Afatinib Axitinib and pembrolizumab Bevacizumab Bosutinib Cabozantinib Cetuximab Crizotinib Dasatinib Erlotinib Everolimus Gefitinib Imatinib Lapatinib with Capecitabine Lenalidomide Nilotinib
Serial No. 1. 2. 3. 4. 5. 6. 7. 8 9. 10. 11. 12. 13. 14. 15.