Page 137 - Read Online
P. 137
Page 10 of 13 Chao et al. Plast Aesthet Res. 2025;12:29 https://dx.doi.org/10.20517/2347-9264.2025.18
indicator is that a neovagina accommodating a finger to the proximal interphalangeal joint (second
knuckle) likely has sufficient length for revision using only peritoneal flaps. This can guide preoperative
counseling and set realistic expectations regarding the need for multiple donor sites.
Intraoperatively, robotic dissection begins in the rectovesical pouch. Placement of a vaginal dilator or end-
to-end anastomosis (EEA) sizer in the remnant canal is helpful. The neovaginal cuff is opened, and
surrounding scar tissue and levator musculature are incised to accommodate the 3.7 cm dilator. In cases of
severe stenosis, the tubularized skin graft is sutured to the introitus using absorbable sutures in a clock-face
pattern and inserted into the vaginal space, easily visualized robotically.
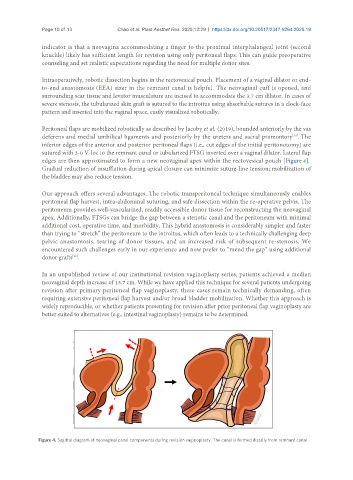
Peritoneal flaps are mobilized robotically as described by Jacoby et al. (2019), bounded anteriorly by the vas
deferens and medial umbilical ligaments and posteriorly by the ureters and sacral promontory . The
[16]
inferior edges of the anterior and posterior peritoneal flaps (i.e., cut edges of the initial peritonotomy) are
sutured with 3-0 V-loc to the remnant canal or tubularized FTSG inverted over a vaginal dilator. Lateral flap
edges are then approximated to form a new neovaginal apex within the rectovesical pouch [Figure 4].
Gradual reduction of insufflation during apical closure can minimize suture-line tension; mobilization of
the bladder may also reduce tension.
Our approach offers several advantages. The robotic transperitoneal technique simultaneously enables
peritoneal flap harvest, intra-abdominal suturing, and safe dissection within the re-operative pelvis. The
peritoneum provides well-vascularized, readily accessible donor tissue for reconstructing the neovaginal
apex. Additionally, FTSGs can bridge the gap between a stenotic canal and the peritoneum with minimal
additional cost, operative time, and morbidity. This hybrid anastomosis is considerably simpler and faster
than trying to “stretch” the peritoneum to the introitus, which often leads to a technically challenging deep
pelvic anastomosis, tearing of donor tissues, and an increased risk of subsequent re-stenosis. We
encountered such challenges early in our experience and now prefer to “mend the gap” using additional
[29]
donor grafts .
In an unpublished review of our institutional revision vaginoplasty series, patients achieved a median
neovaginal depth increase of 13.7 cm. While we have applied this technique for several patients undergoing
revision after primary peritoneal flap vaginoplasty, these cases remain technically demanding, often
requiring extensive peritoneal flap harvest and/or broad bladder mobilization. Whether this approach is
widely reproducible, or whether patients presenting for revision after prior peritoneal flap vaginoplasty are
better suited to alternatives (e.g., intestinal vaginoplasty) remains to be determined.
Figure 4. Sagittal diagram of neovaginal canal components during revision vaginoplasty. The canal is formed distally from remnant canal