Page 114 - Read Online
P. 114
Jiang et al. J Transl Genet Genom 2021;5:323-40 https://dx.doi.org/10.20517/jtgg.2021.21 Page 327
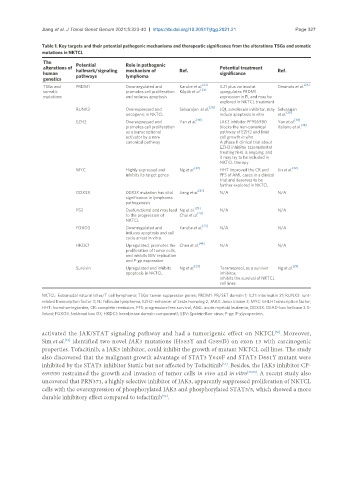
Table 1. Key targets and their potential pathogenic mechanisms and therapeutic significance from the alterations TSGs and somatic
mutations in NKTCL
The Potential Role in pathogenic
alterations of Potential treatment
human hallmark/signaling mechanism of Ref. significance Ref.
pathways
lymphoma
genetics
[23] [25]
TSGs and PRDM1 Downregulated and Karube et al. IL21 plus vorinostat Desmots et al.
somatic promotes cell proliferation Küçük et al. [24] upregulates PRDM1
mutations and reduces apoptosis expression in FL and may be
explored in NKTCL treatment
[30]
RUNX3 Overexpressed and Selvarajan et al. JQ1, a molecule inhibitor, may Selvarajan
oncogenic in NKTCL induce apoptosis in vitro et al. [30]
[36] [36]
EZH2 Overexpressed and Yan et al. JAK3 inhibitor PF956980 Yan et al.
[38]
promotes cell proliferation blocks the non-canonical Italiano et al.
as a transcriptional pathway of EZH2 and limit
activator by a non- cell growth in vitro
canonical pathway A phase II clinical trial about
EZH2 inhibitor tazemetostat
treating NHL is ongoing, and
it may try to be included in
NKTCL therapy
[29] [32]
MYC Highly expressed and Ng et al. HHT improved the CR and Jin et al.
inhibits its target genes PFS of AML cases in a clinical
trial and deserves to be
further explored in NKTCL
[42]
DDX3X DDX3X mutation has vital Jiang et al. N/A N/A
significance in lymphoma
pathogenesis
[29]
P53 Dysfunctional and may lead Ng et al. N/A N/A
to the progression of Choi et al. [43]
NKTCL
FOXO3 Downregulated and Karube et al. [23] N/A N/A
induces apoptosis and cell
cycle arrest in vitro.
[44]
HKDC1 Upregulated, promotes the Chen et al. N/A N/A
proliferation of tumor cells,
and inhibits EBV replication
and P-gp expression.
Survivin Upregulated and inhibits Ng et al. [29] Terameprool, as a survivin Ng et al. [29]
apoptosis in NKTCL. inhibitor,
inhibits the survival of NKTCL
cell lines
NKTCL: Extranodal natural killer/T cell lymphoma; TSGs: tumor suppressor genes; PRDM1: PR/SET domain 1; IL21: interleukin 21; RUNX3: runt-
related transcription factor 3; FL: follicular lymphoma; EZH2: enhancer of zeste homolog 2; JAK3: Janus kinase 3; MYC: bHLH transcription factor;
HHT: homoharringtonine; CR: complete remission; PFS: progression-free survival; AML: acute myeloid leukemia; DDX3X: DEAD-box helicase 3 X-
linked; FOXO3: forkhead box O3; HKDC1: hexokinase domain component1; EBV: Epstein-Barr virus; P-gp: P-glycoprotein.
[50]
activated the JAK/STAT signaling pathway and had a tumorigenic effect on NKTCL . Moreover,
Sim et al. identified two novel JAK3 mutations (H583Y and G589D) on exon 13 with carcinogenic
[51]
properties. Tofacitinib, a JAK3 inhibitor, could inhibit the growth of mutant NKTCL cell lines. The study
also discovered that the malignant growth advantage of STAT3 Y640F and STAT3 D661Y mutant were
inhibited by the STAT3 inhibitor Stattic but not affected by Tofacitinib . Besides, the JAK3 inhibitor CP-
[51]
690550 restrained the growth and invasion of tumor cells in vivo and in vitro [49,50] . A recent study also
uncovered that PRN371, a highly selective inhibitor of JAK3, apparently suppressed proliferation of NKTCL
cells with the overexpression of phosphorylated JAK3 and phosphorylated STAT3/5, which showed a more
durable inhibitory effect compared to tofacitinib .
[52]