Page 298 - Read Online
P. 298
Qureshy et al. J Cancer Metastasis Treat 2020;6:27 I http://dx.doi.org/10.20517/2394-4722.2020.58 Page 3 of 18
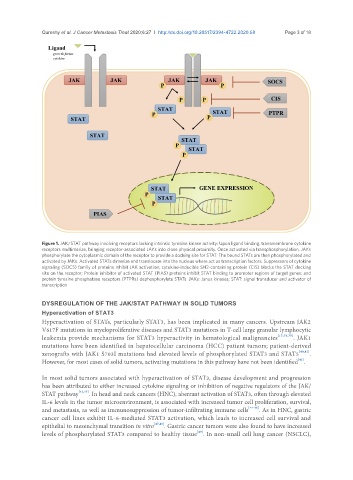
Figure 1. JAK/STAT pathway involving receptors lacking intrinsic tyrosine kinase activity: Upon ligand binding, transmembrane cytokine
receptors multimerize, bringing receptor-associated JAKs into close physical proximity. Once activated via transphosphorylation, JAKs
phosphorylate the cytoplasmic domain of the receptor to provide a docking site for STAT. The bound STATs are then phosphorylated and
activated by JAKs. Activated STATs dimerize and translocate into the nucleus where act as transcription factors. Suppressors of cytokine
signaling (SOCS) family of proteins inhibit JAK activation; cytokine-inducible SH2-containing protein (CIS) blocks the STAT docking
site on the receptor; Protein inhibitor of activated STAT (PIAS) proteins inhibit STAT binding to promoter regions of target genes; and
protein tyrosine phosphatase receptors (PTPRs) dephosphorylate STATs. JAKs: Janus kinases; STAT: signal transducer and activator of
transcription
DYSREGULATION OF THE JAK/STAT PATHWAY IN SOLID TUMORS
Hyperactivation of STAT3
Hyperactivation of STATs, particularly STAT3, has been implicated in many cancers. Upstream JAK2
V617F mutations in myeloproliferative diseases and STAT3 mutations in T-cell large granular lymphocytic
leukemia provide mechanisms for STAT3 hyperactivity in hematological malignancies [15,38,39] . JAK1
mutations have been identified in hepatocellular carcinoma (HCC) patient tumors; patient-derived
xenografts with JAK1 S703I mutations had elevated levels of phosphorylated STAT3 and STAT5 [40,41] .
[42]
However, for most cases of solid tumors, activating mutations in this pathway have not been identified .
In most solid tumors associated with hyperactivation of STAT3, disease development and progression
has been attributed to either increased cytokine signaling or inhibition of negative regulators of the JAK/
STAT pathway [42,43] . In head and neck cancers (HNC), aberrant activation of STAT3, often through elevated
IL-6 levels in the tumor microenvironment, is associated with increased tumor cell proliferation, survival,
and metastasis, as well as immunosuppression of tumor-infiltrating immune cells [44-46] . As in HNC, gastric
cancer cell lines exhibit IL-6-mediated STAT3 activation, which leads to increased cell survival and
epithelial to mesenchymal transition in vitro [47,48] . Gastric cancer tumors were also found to have increased
[49]
levels of phosphorylated STAT3 compared to healthy tissue . In non-small cell lung cancer (NSCLC),