Page 29 - Read Online
P. 29
Miao et al. Energy Mater 2023;3:300014 https://dx.doi.org/10.20517/energymater.2022.89 Page 11 of 31
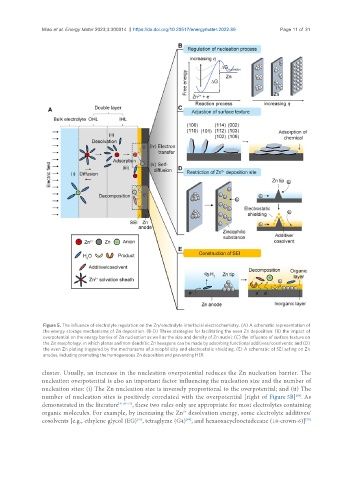
Figure 5. The influence of electrolyte regulation on the Zn/electrolyte interfacial electrochemistry. (A) A schematic representation of
the energy storage mechanisms of Zn deposition. (B-D) Three strategies for facilitating the even Zn deposition: (B) the impact of
overpotential on the energy barrier of Zn nucleation as well as the size and density of Zn nuclei; (C) the influence of surface texture on
the Zn morphology, in which planar and non-dendritic Zn hexagons can be made by adsorbing functional additives/cosolvents; and (D)
the even Zn plating triggered by the mechanisms of zincophilicity and electrostatic shielding. (E) A schematic of SEI acting on Zn
anodes, including promoting the homogeneous Zn deposition and preventing HER.
cluster. Usually, an increase in the nucleation overpotential reduces the Zn nucleation barrier. The
nucleation overpotential is also an important factor influencing the nucleation size and the number of
nucleation sites: (i) The Zn nucleation size is inversely proportional to the overpotential; and (ii) The
number of nucleation sites is positively correlated with the overpotential [right of Figure 5B] . As
[68]
demonstrated in the literature [41,69-71] , these two rules only are appropriate for most electrolytes containing
organic molecules. For example, by increasing the Zn desolvation energy, some electrolyte additives/
2+
[70]
[69]
cosolvents [e.g., ethylene glycol (EG) , tetraglyme (G4) , and hexaoxacyclooctadecane (18-crown-6)]
[41]