Page 138 - Read Online
P. 138
Page 4 of 16 Feng et al. Chem Synth 2023;3:37 https://dx.doi.org/10.20517/cs.2023.26
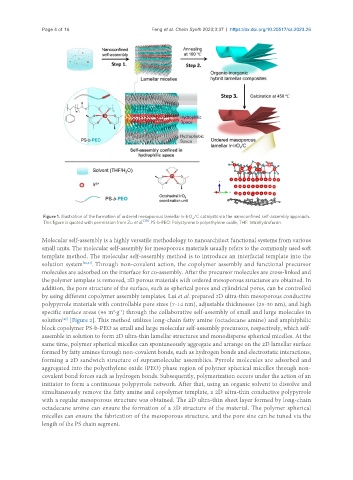
Figure 1. Illustration of the formation of ordered mesoporous lamellar Ir-IrO /C catalysts via the nanoconfined self-assembly approach.
x
This figure is quoted with permission from Zu et al. [39] . PS-b-PEO: Polystyrene b polyethylene oxide; THF: tetrahydrofuran.
Molecular self-assembly is a highly versatile methodology to nanoarchitect functional systems from various
small units. The molecular self-assembly for mesoporous materials usually refers to the commonly used soft
template method. The molecular self-assembly method is to introduce an interfacial template into the
solution system [40,41] . Through non-covalent action, the copolymer assembly and functional precursor
molecules are adsorbed on the interface for co-assembly. After the precursor molecules are cross-linked and
the polymer template is removed, 2D porous materials with ordered mesoporous structures are obtained. In
addition, the pore structure of the surface, such as spherical pores and cylindrical pores, can be controlled
by using different copolymer assembly templates. Lui et al. prepared 2D ultra-thin mesoporous conductive
polypyrrole materials with controllable pore sizes (7-14 nm), adjustable thicknesses (25-30 nm), and high
-1
2
specific surface areas (96 m ·g ) through the collaborative self-assembly of small and large molecules in
solution [Figure 2]. This method utilizes long-chain fatty amine (octadecane amine) and amphiphilic
[42]
block copolymer PS-b-PEO as small and large molecular self-assembly precursors, respectively, which self-
assemble in solution to form 2D ultra-thin lamellar structures and monodisperse spherical micelles. At the
same time, polymer spherical micelles can spontaneously aggregate and arrange on the 2D lamellar surface
formed by fatty amines through non-covalent bonds, such as hydrogen bonds and electrostatic interactions,
forming a 2D sandwich structure of supramolecular assemblies. Pyrrole molecules are adsorbed and
aggregated into the polyethylene oxide (PEO) phase region of polymer spherical micelles through non-
covalent bond forces such as hydrogen bonds. Subsequently, polymerization occurs under the action of an
initiator to form a continuous polypyrrole network. After that, using an organic solvent to dissolve and
simultaneously remove the fatty amine and copolymer template, a 2D ultra-thin conductive polypyrrole
with a regular mesoporous structure was obtained. The 2D ultra-thin sheet layer formed by long-chain
octadecane amine can ensure the formation of a 2D structure of the material. The polymer spherical
micelles can ensure the fabrication of the mesoporous structure, and the pore size can be tuned via the
length of the PS chain segment.