Page 562 - Read Online
P. 562
Page 10 of 13 Maurina et al. Mini-invasive Surg 2021;5:53 https://dx.doi.org/10.20517/2574-1225.2021.88
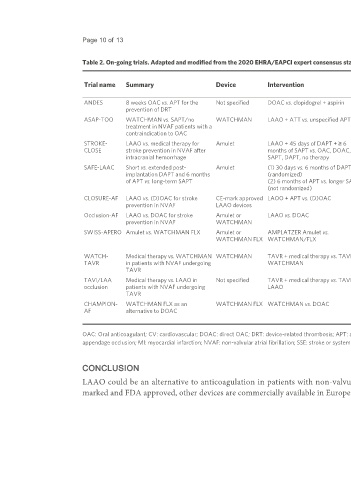
Table 2. On-going trials. Adapted and modified from the 2020 EHRA/EAPCI expert consensus statement update on catheter-based left atrial appendage occlusion [12]
Estimated primary
Patients
Trial name Summary Device Intervention Primary outcome(s) Status and study
enrolled
completion date
ANDES 8 weeks OAC vs. APT for the Not specified DOAC vs. clopidogrel + aspirin 350 2-month DRT Recruiting September 2022 to
prevention of DRT September 2025
ASAP-TOO WATCHMAN vs. SAPT/no WATCHMAN LAAO + ATT vs. unspecified APT 482 7-day device/procedural safety and time to first Active, not December 2025 to
treatment in NVAF patients with a event of SSE recruiting December 2025
contraindication to OAC
STROKE- LAAO vs. medical therapy for Amulet LAAO + 45 days of DAPT + ≥ 6 750 Composite of SSE bleeding and all-cause Recruiting May 2022 to May
CLOSE stroke prevention in NVAF after months of SAPT vs. OAC, DOAC, mortality up to 5 years 2030
intracranial hemorrhage SAPT, DAPT, no therapy
SAFE-LAAC Short vs. extended post- Amulet (1) 30 days vs. 6 months of DAPT 160 Composite of SSE, TIA, non-fatal MI, CV, and Recruiting January 2021 to
implantation DAPT and 6 months (randomized) all-cause mortality, moderate-severe bleeding, January 2022
of APT vs. long-term SAPT (2) 6 months of APT vs. longer SAPT and LAA thrombosis
(not randomized)
CLOSURE-AF LAAO vs. (D)OAC for stroke CE-mark approved LAOO + APT vs. (D)OAC 1512 Survival time free of SSE, major bleeding, and Recruiting February 2021 to
prevention in NVAF LAAO devices CV or unexplained death February 2023
Occlusion-AF LAAO vs. DOAC for stroke Amulet or LAAO vs. DOAC 750 Composite of SSE major bleeding and all-cause Recruiting February 2024 to
prevention in NVAF WATCHMAN mortality up to 5 years October 2030
SWISS-APERO Amulet vs. WATCHMAN FLX Amulet or AMPLATZER Amulet vs. 200 Composite of LAA patency at 45 days and the Active, not July 2021 to May 2026
WATCHMAN FLX WATCHMAN/FLX crossover from one device to the other during recruiting
device implantation
WATCH- Medical therapy vs. WATCHMAN WATCHMAN TAVR + medical therapy vs. TAVR + 350 Composite of all-cause mortality, stroke, and Active, not November 2022 to
TAVR in patients with NVAF undergoing WATCHMAN bleeding recruiting November 2022
TAVR
TAVI/LAA Medical therapy vs. LAAO in Not specified TAVR + medical therapy vs. TAVR + 80 Embolic events, major bleeding, and CV Active, not May 2023 to May
occlusion patients with NVAF undergoing LAAO mortality recruiting 2023
TAVR
CHAMPION- WATCHMAN FLX as an WATCHMAN FLX WATCHMAN vs. DOAC 3000 Non-inferiority for SSE and CV death at 36 Recruiting December 2025 to
AF alternative to DOAC months, non-inferiority for SSE at 60 months, December 2027
and superiority for non-procedural bleeding
OAC: Oral anticoagulant; CV: cardiovascular; DOAC: direct OAC; DRT: device-related thrombosis; APT: antiplatelet therapy; SAPT: single APT; DAPT: double APT; ATT: antithrombotic therapy; LAAO: left atrial
appendage occlusion; MI: myocardial infarction; NVAF: non-valvular atrial fibrillation; SSE: stroke or systemic embolism; TAVR: transcatheter aortic valve replacement.
CONCLUSION
LAAO could be an alternative to anticoagulation in patients with non-valvular AF. While only two devices (WATCHMAN and AMULET) are both CE
marked and FDA approved, other devices are commercially available in Europe, with high procedural success and a low rate of complications.