Page 97 - Read Online
P. 97
Page 8 of 10 de Pascale et al. Mini-invasive Surg 2019;3:18 I http://dx.doi.org/10.20517/2574-1225.2019.04
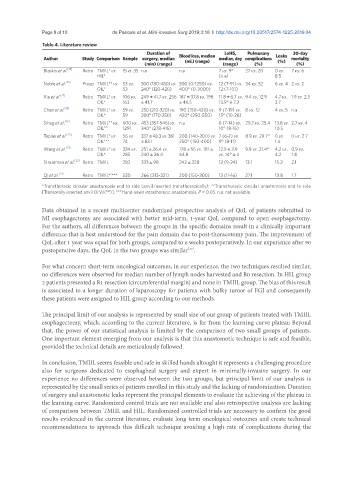
Table 4. Literature review
Duration of Blood loss, median LoHS, Pulmonary Leaks 30-day
Author Study Comparison Sample surgery, median (mL) (range) median, day complications (%) mortality
(min) (range) (range) (%) (%)
Bizekis et al. [15] Retro TMIIL* vs. 15 vs. 35 n.a n.a 7 vs. 9° 27 vs. 20 0 vs. 7 vs. 6
HIL* (n.a) 8.5
Noble et al. [16] Prosp TMIIL** vs. 53 vs. 300 (180-480) vs. 300 (0-1250) vs. 12 (7-91) vs. 34 vs. 32 6 vs. 4 2 vs. 2
OIL* 53 240° (120-420) 400° (0-3000) 12 (7-101)
Xie et al. [17] Retro TMIIL* vs. 106 vs. 249 ± 41.7 vs. 256 187 ± 37.8 vs. 198 11.8 ± 6.7 vs. 9.4 vs. 12.9 4.7 vs. 1.9 vs. 2.5
OIL* 163 ± 41.7 ± 46.5 13.9° ± 7.3 3.7
Chen et al. [18] Retro TMIIL* vs. 59 vs. 250 (210-320) vs. 190 (150-420) vs. 9 (7-19) vs. 8 vs. 12 4 vs. 5 n.a
OIL* 59 200° (170-250) 420° (250-550) 15° (10-28)
n.a
Sihag et al. [19] Retro TMIIL vs. 600 vs. 453 (357-546) vs. n.a 8 (7-14) vs. 29.7 vs. 25.4 13.8 vs. 2.7 vs. 4
OIL n.a 1291 340° (278-415) 10° (8-16) 10.5
Tapias et al. [20] Retro TMIIL* vs. 56 vs. 337 ± 48.3 vs. 361 200 (140-200) vs. 7 (6-7) vs. 8.9 vs. 29.7° 0 vs. 0 vs. 2.7
OIL*** 74 ± 83.1 250° (150-400) 9° (8-11) 1.4
Wang et al. [21] Retro TMIIL* vs. 334 vs. 251 ± 26.4 vs. 178 ± 55 vs. 181 ± 12.9 ± 3.9 9.9 vs. 21.4° 4.2 vs. 0.9 vs.
OIL* 285 240 ± 26.4 64.8 vs. 14° ± 4 4.2 1.4
Straatman et al. [22] Retro TMIIL 282 333 ± 98 242 ± 228 12 (9-24) 13.1 15.2 2.1
/
Qi et al. [23] Retro TMIIL* *** 530 266 (213-321) 200 (150-300) 13 (11-16) 27.1 13.8 1.7
*Transthoracic circular anastomosis end to side (anvil inserted transthoracically); **Transthoracic circular anastomosis end to side
[Transorally inserted anvil OrVil(™)]; ***Hand-sewn intrathoracic anastomosis, P < 0.05. n.a: not available
Data obtained in a recent multicenter randomized prospective analysis of QoL of patients submitted to
MI esophagectomy are associated with better mid-term, 1-year QoL compared to open esophagectomy.
For the authors, all differences between the groups in the specific domains result in a clinically important
difference that is best understood for the pain domain due to post-thoracotomy pain. The improvement of
QoL after 1 year was equal for both groups, compared to 6 weeks postoperatively. In our experience after 90
postoperative days, the QoL in the two groups was similar .
[27]
For what concern short-term oncological outcomes, in our experience, the two techniques resulted similar,
no differences were observed for median number of lymph nodes harvested and R0 resection. In HIL group
2 patients presented a R1 resection (circumferential margin) and none in TMIIL group. The bias of this result
is associated to a longer duration of laparoscopy for patients with bulky tumor of EGJ and consequently
these patients were assigned to HIL group according to our methods.
The principal limit of our analysis is represented by small size of our group of patients treated with TMIIL
esophagectomy, which, according to the current literature, is far from the learning curve plateau Beyond
that, the power of our statistical analysis is limited by the comparison of two small groups of patients.
One important element emerging from our analysis is that this anastomotic technique is safe and feasible,
provided the technical details are meticulously followed.
In conclusion, TMIIL seems feasible and safe in skilled hands altought it represents a challenging procedure
also for surgeons dedicated to esophagheal surgery and expert in minimally-invasive surgery. In our
experience no differences were observed between the two groups, but principal limit of our analysis is
represented by the small series of patients enrolled in this study and the lacking of randomization. Duration
of surgery and anastomotic leaks represent the principal elements to evaluate the achieving of the plateau in
the learning curve. Randomized control trials are not available and also retrospective analysis are lacking
of comparison between TMIIL and HIL. Randomized controlled trials are necessary to confirm the good
results evidenced in the current literature, evaluate long term oncological outcomes and create technical
recommendations to approach this difficult technique avoiding a high rate of complications during the