Page 56 - Read Online
P. 56
Author Instructions
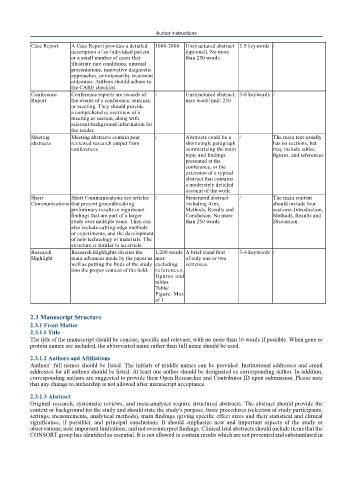
Case Report A Case Report provides a detailed 1000-2000 Unstructured abstract 2-5 keywords /
description of an individual patient (optional). No more
or a small number of cases that than 250 words.
illustrate rare conditions, unusual
presentations, innovative diagnostic
approaches, or noteworthy treatment
outcomes. Authors should adhere to
the CARE checklist.
Conference Conference reports are records of / Unstructured abstract; 3-8 keywords /
Report the events of a conference, seminar, max word limit: 250
or meeting. They should provide
a comprehensive overview of a
meeting or session, along with
relevant background information for
the reader.
Meeting Meeting abstracts contain peer / Abstracts could be a / The main text usually
abstracts reviewed research output from short single paragraph has no sections, but
conferences summarizing the main may include tables,
topic and findings figures, and references.
presented at the
conference, or the
extension of a typical
abstract that contains
a moderately detailed
account of the work.
Short Short Communications are articles / Structured abstract / The main content
Communications that present groundbreaking including Aim, should include four
preliminary results or significant Methods, Results and sections: Introduction,
findings that are part of a larger Conclusion. No more Methods, Results and
study over multiple years. They can than 250 words Discussion.
also include cutting-edge methods
or experiments, and the development
of new technology or materials. The
structure is similar to an article.
Research Research Highlights discuss the 1,200 words A brief stand first 3-6 keywords /
Highlight main advances made by the paper as max of only one or two
well as putting the finds of the study excluding, sentences.
into the proper context of the field. references,
figures and
tables.
Table/
Figure: Max
of 1
2.3 Manuscript Structure
2.3.1 Front Matter
2.3.1.1 Title
The title of the manuscript should be concise, specific and relevant, with no more than 16 words if possible. When gene or
protein names are included, the abbreviated name rather than full name should be used.
2.3.1.2 Authors and Affiliations
Authors’ full names should be listed. The initials of middle names can be provided. Institutional addresses and email
addresses for all authors should be listed. At least one author should be designated as corresponding author. In addition,
corresponding authors are suggested to provide their Open Researcher and Contributor ID upon submission. Please note
that any change to authorship is not allowed after manuscript acceptance.
2.3.1.3 Abstract
Original research, systematic reviews, and meta-analyses require structured abstracts. The abstract should provide the
context or background for the study and should state the study’s purpose, basic procedures (selection of study participants,
settings, measurements, analytical methods), main findings (giving specific effect sizes and their statistical and clinical
significance, if possible), and principal conclusions. It should emphasize new and important aspects of the study or
observations, note important limitations, and not overinterpret findings. Clinical trial abstracts should include items that the
CONSORT group has identified as essential. It is not allowed to contain results which are not presented and substantiated in