Page 155 - Read Online
P. 155
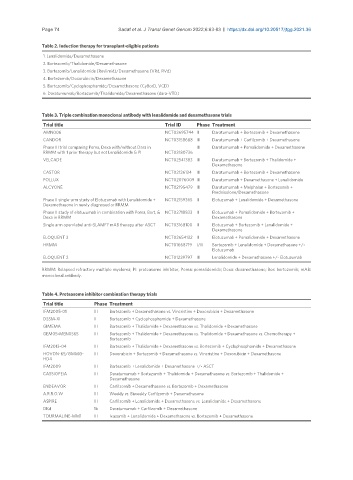
Page 74 Sadaf et al. J Transl Genet Genom 2022;6:63-83 https://dx.doi.org/10.20517/jtgg.2021.36
Table 2. Induction therapy for transplant-eligible patients
1. Lenalidomide/Dexamethasone
2. Bortezomib/Thalidomide/Dexamethasone
3. Bortezomib/Lenalidomide (Revlimid)/Dexamethasone (VRd, RVd)
4. Bortezomib/Doxorubicin/Dexamethasone
5. Bortezomib/Cyclophosphamide/Dexamethasone (CyBorD, VCD)
6. Daratumumab/Bortezomib/Thalidomide/Dexamethasone (dara-VTD)
Table 3. Triple combination monoclonal antibody with lenalidomide and dexamethasone trials
Trial title Trial ID Phase Treatment
AMN006 NCT03695744 II Daratumumab + Bortezomib + Dexamethasone
CANDOR NCT03158688 III Daratumumab + Carfilzomib + Dexamethasone
Phase III trial comparing Poma, Dexa with/without Dara in III Daratumumab + Pomalidomide + Dexamethasone
RRMM with 1 prior therapy but not Lenalidomide & PI NCT03180736
VELCADE NCT02541383 III Daratumumab + Bortezomib + Thalidomide +
Dexamethasone
CASTOR NCT02136134 III Daratumumab + Bortezomib + Dexamethasone
POLLUX NCT02076009 III Daratumumab + Dexamethasone + Lenalidomide
ALCYONE NCT02195479 III Daratumumab + Melphalan + Bortezomib +
Prednisolone/Dexamethasone
Phase II single-arm study of Elotuzumab with Lenalidomide + NCT02159365 II Elotuzumab + Lenalidomide + Dexamethasone
Dexamethasone in newly diagnosed or RRMM
Phase II study of elotuzumab in combination with Poma, Bort, & NCT02718833 II Elotuzumab + Pomalidomide + Bortezomib +
Dexa in RRMM Dexamethasone
Single arm open-label anti-SLAMF7 mAB therapy after ASCT NCT03168100 II Elotuzumab + Bortezomib + Lenalidomide +
Dexamethasone
ELOQUENT 3 NCT02654132 II Elotuzumab + Pomalidomide + Dexamethasone
HRMM NCT01668719 I/II Bortezomib + Lenalidomide + Dexamethasone +/-
Elotuzumab
ELOQUENT 2 NCT01239797 III Lenalidomide + Dexamethasone +/- Elotuzumab
RRMM: Relapsed refractory multiple myeloma; PI: proteasome inhibitor, Poma: pomalidomide; Dexa: dexamethasone; Bor: bortezomib; mAB:
monoclonal antibody.
Table 4. Proteasome inhibitor combination therapy trials
Trial title Phase Treatment
IFM2005-01 III Bortezomib + Dexamethasone vs. Vincristine + Doxorubicin + Dexamethasone
DSSM-XI II Bortezomib + Cyclophosphamide + Dexamethasone
GIMEMA III Bortezomib + Thalidomide + Dexamethasone vs. Thalidomide + Dexamethasone
GEM05-MEN0S65 III Bortezomib + Thalidomide + Dexamethasone vs. Thalidomide + Dexamethasone vs. Chemotherapy +
Bortezomib
IFM2013-04 III Bortezomib + Thalidomide + Dexamethasone vs. Bortezomib + Cyclophosphamide + Dexamethasone
HOVON-65/GMMG- III Doxorubicin + Bortezomib + Dexamethasone vs. Vincristine + Doxorubicin + Dexamethasone
HD4
IFM2009 III Bortezomib + Lenalidomide + Dexamethasone +/- ASCT
CASSIOPEIA III Daratumumab + Bortezomib + Thalidomide + Dexamethasone vs. Bortezomib + Thalidomide +
Dexamethasone
ENDEAVOR III Carfilzomib + Dexamethasone vs. Bortezomib + Dexamethasone
A.R.R.O.W III Weekly vs. Biweekly Carfilzomib + Dexamethasone
ASPIRE III Carfilzomib + Lenalidomide + Dexamethasone vs. Lenalidomide + Dexamethasone
DKd 1b Daratumumab + Carfilzomib + Dexamethasone
TOURMALINE-MM1 III Ixazomib + Lenalidomide + Dexamethasone vs. Bortezomib + Dexamethasone