Page 122 - Read Online
P. 122
Page 8 of 10 Simsek et al. Hepatoma Res 2020;6:11 I http://dx.doi.org/10.20517/2394-5079.2019.51
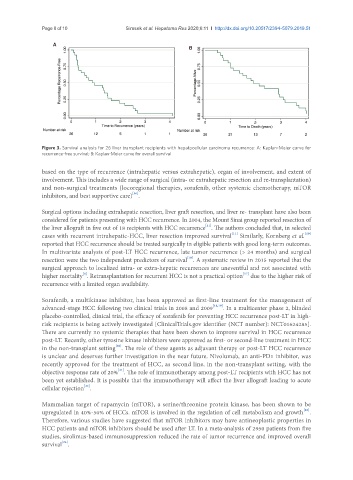
A
B
Figure 3. Survival analysis for 26 liver transplant recipients with hepatocellular carcinoma recurrence: A: Kaplan-Meier curve for
recurrence-free survival; B: Kaplan-Meier curve for overall survival
based on the type of recurrence (intrahepatic versus extrahepatic), organ of involvement, and extent of
involvement. This includes a wide range of surgical (intra- or extrahepatic resection and re-transplantation)
and non-surgical treatments (locoregional therapies, sorafenib, other systemic chemotherapy, mTOR
[16]
inhibitors, and best supportive care) .
Surgical options including extrahepatic resection, liver graft resection, and liver re- transplant have also been
considered for patients presenting with HCC recurrence. In 2004, the Mount Sinai group reported resection of
[11]
the liver allograft in five out of 18 recipients with HCC recurrence . The authors concluded that, in selected
cases with recurrent intrahepatic-HCC, liver resection improved survival Similarly, Kornberg et al.
[11]
[10]
reported that HCC recurrence should be treated surgically in eligible patients with good long-term outcomes.
In multivariate analysis of post-LT HCC recurrence, late tumor recurrence (> 24 months) and surgical
[10]
resection were the two independent predictors of survival . A systematic review in 2015 reported that the
surgical approach to localized intra- or extra-hepatic recurrences are uneventful and not associated with
[8]
[17]
higher mortality . Retransplantation for recurrent HCC is not a practical option due to the higher risk of
recurrence with a limited organ availability.
Sorafenib, a multikinase inhibitor, has been approved as first-line treatment for the management of
advanced-stage HCC following two clinical trials in 2008 and 2009 [18,19] . In a multicenter phase 2, blinded
placebo-controlled, clinical trial, the efficacy of sorafenib for preventing HCC recurrence post-LT in high-
risk recipients is being actively investigated [ClinicalTrials.gov identifier (NCT number): NCT01624285].
There are currently no systemic therapies that have been shown to improve survival in HCC recurrence
post-LT. Recently, other tyrosine kinase inhibitors were approved as first- or second-line treatment in HCC
in the non-transplant setting . The role of these agents as adjuvant therapy or post-LT HCC recurrence
[20]
is unclear and deserves further investigation in the near future. Nivolumab, an anti-PD1 inhibitor, was
recently approved for the treatment of HCC, as second line, in the non-transplant setting, with the
[21]
objective response rate of 20% . The role of immunotherapy among post-LT recipients with HCC has not
been yet established. It is possible that the immunotherapy will affect the liver allograft leading to acute
[22]
cellular rejection .
Mammalian target of rapamycin (mTOR), a serine/threonine protein kinase, has been shown to be
[23]
upregulated in 40%-50% of HCCs. mTOR is involved in the regulation of cell metabolism and growth .
Therefore, various studies have suggested that mTOR inhibitors may have antineoplastic properties in
HCC patients and mTOR inhibitors should be used after LT. In a meta-analysis of 2950 patients from five
studies, sirolimus-based immunosuppression reduced the rate of tumor recurrence and improved overall
survival .
[24]