Page 973 - Read Online
P. 973
Page 6 of 12 Lonardo et al. Hepatoma Res 2020;6:83 I http://dx.doi.org/10.20517/2394-5079.2020.89
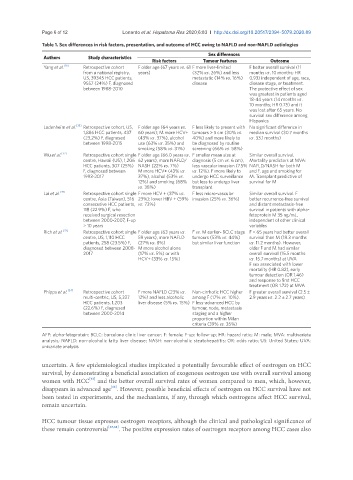
Table 1. Sex differences in risk factors, presentation, and outcome of HCC owing to NAFLD and non-NAFLD aetiologies
Sex differences
Authors Study characteristics
Risk factors Tumour features Outcome
Yang et al. [16] Retrospective cohort F older age (67 years vs. 61 F more liver-limited F better overall survival (11
from a national registry, years) (32% vs. 26%) and less months vs. 10 months; HR
US, 39345 HCC patients, metastatic (14% vs. 16%) 0.93) independent of age, race,
9557 (24%) F, diagnosed disease disease stage, or treatment.
between 1988-2010 The protective effect of sex
was greatest in patients aged
18-44 years (14 months vs.
10 months; HR 0.75) and it
was lost after 65 years. No
survival sex difference among
Hispanics
Ladenheim et al. [35] Retrospective cohort, US, F older age (64 years vs. F less likely to present with No significant difference in
1,886 HCC patients, 437 60 years); M more HCV+ tumours > 5 cm (30% vs. median survival (30.7 months
(23.2%) F, diagnosed (43% vs. 37%), alcohol 40%) and more likely to vs. 33.1 months)
between 1998-2015 use (63% vs. 35%) and be diagnosed by routine
smoking (58% vs. 31%) screening (66% vs. 58%)
Wu et al. [32] Retrospective cohort single F older age (66.0 years vs. F smaller mean size at Similar overall survival.
centre, Hawaii (US), 1,206 62 years), more NAFLD/ diagnosis (5 cm vs. 6 cm), Mortality predictors at MVA:
HCC patients, 307 (25%) NASH (22% vs. 7%) less vascular invasion (7.5% NAFLD/NASH for both M
F, diagnosed between M more HCV+ (43% vs. vs. 12%). F more likely to and F, age and smoking for
1993-2017 37%), alcohol (53% vs. undergo HCC surveillance M. Transplant predictive of
12%) and smoking (68% but less to undergo liver survival for M
vs. 38%) transplant
Lai et al. [39] Retrospective cohort single F more HCV + (37% vs. F less micro-vascular Similar overall survival. F
centre, Asia (Taiwan), 516 23%); lower HBV + (59% invasion (25% vs. 36%) better recurrence-free survival
consecutive HCC patients, vs. 73%) and distant metastasis-free
118 (22.9%) F, who survival in patients with alpha-
received surgical resection fetoprotein ≤ 35 ng/mL,
between 2000-2007, F-up independent of other clinical
> 10 years variables
Rich et al. [33] Retrospective cohort single F older age (63 years vs. F vs. M earlier- BCLC stage F < 65 years had better overall
centre, US, 1,110 HCC 59 years), more NAFLD tumours (53% vs. 44%) survival than M (18.3 months
patients, 258 (23.5%) F, (27% vs. 8%) but similar liver function vs. 11.2 months). However,
diagnosed between 2008- M more alcohol alone older F and M had similar
2017 (17% vs. 5%) or with overall survival (15.5 months
HCV+ (33% vs. 15%) vs. 15.7 months) at UVA
F sex associated with lower
mortality (HR 0.82), early
tumour detection (OR 1.46)
and response to first HCC
treatment (OR 1.72) at MVA
Phipps et al. [34] Retrospective cohort F more NAFLD (23% vs. Non-cirrhotic HCC higher F greater overall survival (2.5 ±
multi-centric, US, 5,327 12%) and less alcoholic among F (17% vs. 10%). 2.9 years vs. 2.2 ± 2.7 years)
HCC patients, 1,203 liver disease (5% vs. 15%) F less-advanced HCC by
(22.6%) F, diagnosed tumour, node, metastasis
between 2000-2014 staging and a higher
proportion within Milan
criteria (39% vs. 35%)
AFP: alpha-fetoprotein; BCLC: barcelona clinic liver cancer; F: female; F-up: follow-up; HR: hazard ratio; M: male; MVA: multivariate
analysis; NAFLD: non-alcoholic fatty liver disease; NASH: non-alcoholic steatohepatitis; OR: odds ratio; US: United States; UVA:
univariate analysis
uncertain. A few epidemiological studies implicated a potentially favourable effect of oestrogen on HCC
survival, by demonstrating a beneficial association of exogenous oestrogen use with overall survival among
[62]
women with HCC and the better overall survival rates of women compared to men, which, however,
[16]
disappears in advanced age . However, possible beneficial effects of oestrogen on HCC survival have not
been tested in experiments, and the mechanisms, if any, through which oestrogens affect HCC survival,
remain uncertain.
HCC tumour tissue expresses oestrogen receptors, although the clinical and pathological significance of
these remain controversial [63,64] . The positive expression rates of oestrogen receptors among HCC cases also